Newer MRI-guided focused ultrasound (MRgFUS) systems work better for successfully treating uterine fibroids, according to a July 4 study in Academic Radiology. A second-generation system significantly reduced fibroid volume while lowering the percentage of patients with complications.
MRgFUS is a minimally invasive treatment for uterine fibroids that uses ultrasound acoustic energy to thermally ablate tissue from outside of the body. Newer systems benefit from a number of modifications, including a higher maximum energy level and closer transducer placement, the authors noted.
"This study has demonstrated that improvements to current MRgFUS technology resulted in significantly increased efficacy and patient safety of clinical treatments of patients with symptomatic uterine leiomyomas," wrote the authors, led by Jacinta Browne, PhD, from the Mayo Clinic department of radiology.
The U.S. Food and Drug Administration (FDA) has approved two MRgFUS systems for uterine fibroid treatment: InSightec's ExAblate 2000 and ExAblate 2100. The systems received approval in 2004 and 2009, respectively.
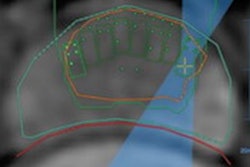
The 2100 model features a number of upgrades over its predecessor, including a new 3D axis ultrasound transducer with movement capabilities, the ability to shut off the transducers near high-risk structures, and a higher maximum sonication energy level of 7,200 J.
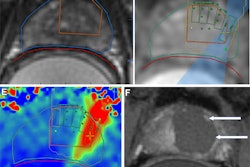
The authors compared the efficacy of the two MRgFUS models on patients treated for symptomatic uterine fibroids at the Mayo Clinic in Rochester, MN, between 2005 and 2019. The first group of 136 patients was treated with the original ExAblate system, while the second group of 73 patients was treated with the newer model.
The ExAblate 2100 group had a treatment completion rate of 97.3%, compared to 95.6% in the ExAblate 2000 group. In the group treated with the original model, treatment was not completed for multiple reasons, including insufficient temperature, fibroid position, or anatomical constraints. Whereas insufficient temperature was the only reason for not completing treatment with the newer model.
Furthermore, fewer patients developed complications with the second-generation system. In the ExAblate 2000 group, 13% experienced complications, including mild abdominal edema, treatment-associated lower back discomfort, and deep vein thrombosis. But only 2 patients (5.4%) in the ExAblate 2100 group developed complications, and both complications were mild abdominal edema that resolved on its own.
The findings were even more substantial when the authors specifically looked at the percentage of women who achieved a nonperfused fibroid volume rate of at least 60%, a threshold commonly used to demarcate treatment success. At this threshold, 86% of patients in the ExAblate 2100 group had successful treatment, compared with just 19% of patients in the ExAblate 2000 group.
"In our opinion, this improvement was largely due to the major technical modifications which were implemented on the ExAblate 2100 aimed at compensating for both technical and anatomical limitations previously restricting access to all fibroid tissues due to anatomical locations," the authors wrote.
Although the study only included immediate after-treatment results, the findings still showed the improved clinical ability of the second-generation MRgFUS system. The authors also intend to conduct follow-up questionnaires with all study participants.
"This study has demonstrated that fibroid treatments carried out using the second-generation ExAblate 2100 MRgFUS system, which utilized technological advancements, had a significant improvement in the initial treatment outcomes compared to the first generation ExAblate 2000 system," the authors concluded.