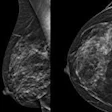
The U.S. Food and Drug Administration (FDA) is proposing to amend its regulations governing mammography to include breast density reporting, the agency stated in the Federal Register.
The FDA is proposing to add a breast density reporting amendment to the Mammography Quality Standards Act (MQSA), which would mean that women would receive standardized nationwide notification about their breast tissue density after their mammograms. The FDA said it would consider the proposed rule in December 2013.
In addition to breast density notification, the amendments would update MQSA regulations to address changes in mammography technology and processes that have occurred since the regulations were last published in 1997.