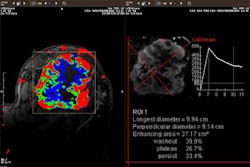
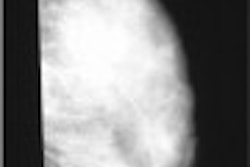
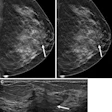
CT laser mammography developer Imaging Diagnostic Systems has been notified by the Food and Drug Administration that an initial review of its pre-market approval (PMA) application for its CT breast imaging system has been judged to be suitable for filing. As such, the FDA will now conduct an in-depth evaluation of the safety and effectiveness of the device to determine final approval, according to the Fort Lauderdale, FL-based vendor.
By AuntMinnie.com staff writersJune 18, 2003
Related Reading
Imaging Diagnostic Systems receives 12th U.S. patent, May 29, 2003
Imaging Diagnostic Systems files PMA, April 30, 2003
Imaging Diagnostic Systems adds Berlin site, February 3, 2003
IMDS nets CTLM deal in Dubai, January 23, 2003
IDSI installs CTLM in Berlin, September 12, 2002
Copyright © 2003 AuntMinnie.com