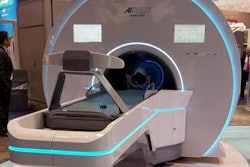
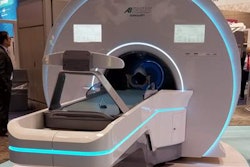
Radiation therapy developer Akesis has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its Akesis Galaxy RTx radiosurgery system.
The Akesis Galaxy RTx is a new gamma stereotactic radiosurgery system that's based on Akesis Galaxy.
The RTx model incorporates 30 gamma sources into a rotational design. The configuration gives users more options to shape dose distribution versus traditional, fixed-sector radiation delivery, according to Akesis.












![A 53-year-old patient (patient number four) with a recurrent pituitary adenoma with extension of a cystic component of disease to the medial temporal lobe apparent on MRI (contoured in blue), and extension of disease to the left sphenoid bone and orbital apex apparent on [68Ga]Ga-DOTA-TATE (contoured in yellow).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/04/pituitary-tumor.QGsEnyB4bU.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)



