Higher doses in external beam radiotherapy (RT) for localized prostate cancer improve patient outcomes with lower prostate-specific antigen recurrence rates, but rectal toxicity is one of the dangers of this approach.
Further complicating matters, the natural motion of the prostate (day-to-day gland displacements range from 3 mm to 5 mm) can make pinpointing RT treatment difficult. To make treatment margins as accurate as possible, radiopaque markers are implanted in the prostate to aid in localizing the gland during RT. But are these markers safe?
The use of gold markers is increasingly common, according to Dutch researchers. But not much data on complication rates in large patient populations have been reported. Dr. Johan Langenhuijsen and colleagues at the Radboud University Nijmegen Medical Centre in Nijmegen, Netherlands, reported results from their study analyzing complication rates and identifying the risk factors for the complications in patients with localized prostate cancer who had received gold markers transrectally (International Journal of Radiation Oncology, Biology, Physics, November 2007, Vol. 69: 3, pp 671-676).
The study, which occurred between January 2001 and September 2005, included 209 men with localized prostate cancer who had been referred for external beam RT. Of these 209 men, the tumor was stage T1 in 18, T2 in 64, and T3 in 127. The mean age was 70, and the age range was 40 to 84 years.
Four gold markers, each 1 x 7 mm, were inserted with ultrasound guidance in an outpatient setting. All patients received a questionnaire with which to track complications after the procedure; complications and risk factors were further assessed by reviewing each participant's medical charts. None of the men received enemas or anesthesia before the procedure; all received a prophylactic antibiotic for three days. Any participants on anticoagulant medication discontinued its use for three to seven days before the markers were implanted. Hormonal treatment was begun in 202 participants with a mean interval of seven weeks until the procedure was performed. The mean interval between implantation of the markers and the start of RT was 26 days.
The gold markers were placed with an 18-guage prostate biopsy tool mounted onto the endorectal ultrasound transducer. Two markers were placed to the left and right of the base of the gland, one on the central part next to the urethra, and one at the top of the prostate. After one week, to allow for any swelling after implementation to resolve, an RT planning CT scan was performed.
With a take-home questionnaire, participants were asked to evaluate the daily frequency and duration of the following symptoms: blood in the urine or seminal fluid, rectal bleeding, fever, and pain. Pain was scored on a scale of 0 to 10 (0 being no pain, 10 being worst pain imaginable).
|
Study summary Of the 209 men included in the study:
|
The mean pain score was 3.2, with a range of 0 to 9. Of the 209 men included in the study, 48% scored the pain as 0 to 2, 37% as 3 to 5, and 15% as 6 to 9. Fifty percent of those included in the study reported that the marker implantation procedure was less painful than the prostate biopsy procedure.
As for moderate complications, 6.2% experienced pain and fever, 1.9% experienced minor voiding complaints such as dysuria, 9.1% experienced rectal bleeding, 3.8% experienced blood in urine lasting more than three days, 18.5% experienced blood in seminal fluid (in 15 of 81 participants reporting ejaculations), and 9.1% experienced rectal bleeding.
Although the risk of bleeding complications after the marker implantation procedure was higher for the men with tumors in advanced stages, those who were younger in age, and those who had undergone a shorter duration of hormonal treatment, Langenhuijsen and colleagues concluded that transrectal gold marker implantation for prostate position verification in RT is safe and well-tolerated.
By Kate Madden Yee
AuntMinnie.com staff writer
November 20, 2007
Related Reading
Previous prostate radiation therapy linked to higher-grade bladder cancer, July 18, 2006
Focal cryoablation promoted as new paradigm for treating prostate cancer, April 4, 2005
Prostate cancer family history does not predict benign hyperplasia, April 4, 2005
Prostate radiotherapy raises risk of rectal cancer, April 1, 2005<
MR method assesses prostate cancer aggressiveness noninvasively, March 7, 2005
Copyright © 2007 AuntMinnie.com




![A 53-year-old patient (patient number four) with a recurrent pituitary adenoma with extension of a cystic component of disease to the medial temporal lobe apparent on MRI (contoured in blue), and extension of disease to the left sphenoid bone and orbital apex apparent on [68Ga]Ga-DOTA-TATE (contoured in yellow).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/04/pituitary-tumor.QGsEnyB4bU.jpg?auto=format%2Ccompress&fit=crop&h=100&q=70&w=100)

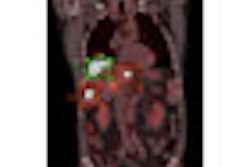





![A 53-year-old patient (patient number four) with a recurrent pituitary adenoma with extension of a cystic component of disease to the medial temporal lobe apparent on MRI (contoured in blue), and extension of disease to the left sphenoid bone and orbital apex apparent on [68Ga]Ga-DOTA-TATE (contoured in yellow).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/04/pituitary-tumor.QGsEnyB4bU.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)