The Radiation Management Services unit of Fluke Biomedical has received the European CE Mark for its Double Check Pro Model 7600, a portable device for therapy beam quality assurance.
The standalone product allows for treatment beam data analysis, including flatness, symmetry, constancy, and chart and graphical data presentation. It also features removable and internal flash memory and Ethernet, USB, and wireless communication capabilities, according to the Cleveland-based firm.
By AuntMinnie.com staff writers
June 23, 2005
Related Reading
Fluke debuts treatment brassiere, June 14, 2005
Fluke unit launches monitor calibration tool, May 12, 2005
Fluke debuts nested CT dose phantom set, April 20, 2005
Cardinal sells RMS business, January 6, 2005
Copyright © 2005 AuntMinnie.com
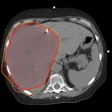
![A 53-year-old patient (patient number four) with a recurrent pituitary adenoma with extension of a cystic component of disease to the medial temporal lobe apparent on MRI (contoured in blue), and extension of disease to the left sphenoid bone and orbital apex apparent on [68Ga]Ga-DOTA-TATE (contoured in yellow).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/04/pituitary-tumor.QGsEnyB4bU.jpg?auto=format%2Ccompress&fit=crop&h=100&q=70&w=100)




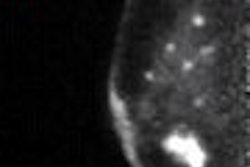
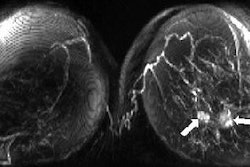

![A 53-year-old patient (patient number four) with a recurrent pituitary adenoma with extension of a cystic component of disease to the medial temporal lobe apparent on MRI (contoured in blue), and extension of disease to the left sphenoid bone and orbital apex apparent on [68Ga]Ga-DOTA-TATE (contoured in yellow).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/04/pituitary-tumor.QGsEnyB4bU.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)