In results they described as "unexpected" and "controversial," Norwegian researchers found levels of residual signal enhancement in the brains of 17 patients who were given a macrocyclic gadolinium-based contrast agent (GBCA), with the highest levels found in two patients who received the most doses, according to a paper in the November issue of Radiology.
The findings contradict several previous studies in which multiple injections of macrocyclic GBCAs showed no evidence of measurable gadolinium retention. In the current study, the researchers saw the highest levels of residual gadolinium in the dentate nucleus of two patients who had received 37 and 44 standard administrations, respectively, of gadobutrol (Gadavist, Bayer HealthCare) (Radiology, November 2017, Vol. 285:2, pp. 434-444).
"Our results were unexpected and are likely controversial," wrote lead author Atle Bjørnerud, PhD, and colleagues from Oslo University Hospital. "These results raise the question whether visible enhancement based on standard radiologic reading is an absolute criterion to confirm gadolinium deposition in the dentate nucleus or whether optimized image contrast or region-of-interest measurements may reveal subtle enhancement effects not evident with standard visual inspection."
The macrocyclic effect
For several years now, studies from around the world have confirmed minute traces of gadolinium in the tissue of patients long after they received contrast-enhanced MRI scans. Researchers also explored how linear and macrocyclic GBCAs produce different levels of signal intensity in the dentate nucleus.
Late last year, Radbruch et al found no evidence of increases in T1 signal intensity among patients who received more than 20 injections of two commercially available macrocyclic GBCAs: gadobutrol and gadoterate meglumine (Dotarem, Guerbet). The researchers also concluded that switching from a linear GBCA to a macrocyclic GBCA reduced the level of residual signal intensity in the brains of patients who underwent multiple enhanced MRI scans with both types of agents.
Earlier this year, McDonald et al confirmed in a prospective animal study the presence of gadolinium deposits in brain tissue after the injection of both linear and macrocyclic GBCAs. That study compared two linear GBCAs -- gadodiamide (Omniscan, GE Healthcare) and gadobenate dimeglumine (MultiHance, Bracco Imaging) -- with two macrocyclic GBCAs -- gadobutrol and gadoteridol (ProHance, Bracco).
In the current study, Bjørnerud and colleagues decided they wanted to research whether signal enhancement occurred in the dentate nucleus in patients who had no history of linear GBCA injections but who had received a large number of macrocyclic GBCA injections, resulting in a large range of gadolinium dose.
They retrospectively reviewed 17 patients with high-grade gliomas who had received 10 to 44 administrations of the macrocyclic GBCA gadobutrol (0.1 mmol/kg of body weight) for exams on a 3-tesla MRI system (Achieva, Philips Healthcare). The mean number of administrations was 22.2 (± 10.6) over a mean of 706 days (± 454 days).
The MRI protocol for these subjects included structural imaging with high-spatial-resolution 2D and 3D T2-weighted imaging, unenhanced and gadobutrol-enhanced 3D T1-weighted imaging, diffusion-tensor imaging, and gadobutrol-enhanced T1- and T2-weighted dynamic perfusion.
Two neuroradiologists then analyzed T1-weighted MR images to evaluate any differences between baseline scans and the last exam in terms of visual dentate nucleus signal enhancement.
More specifically, the duo rated signal intensity separately for the left and right dentate nucleus, with 0 as no enhancement, 1 as slight enhancement, and 2 as strong enhancement. The neuroradiologists also rated whether enhancement was confined to the dentate nucleus (1) or it went beyond the dentate nucleus (2).
Signal enhancement
In reviewing images of the 17 patients, the researchers found statistically significant and dose-dependent increases in dentate nucleus signal intensity between the last MRI and baseline exams. Mean signal intensity from the last MRI scan was greater than 0 (0.03 ± 0.05; p = 0.016).
In addition, the difference in signal intensity between the last MRI scan (r = 0.69, p = 0.002) and the baseline signal intensity (r = 0.77) varied based on the number of macrocyclic GBCA injections the patient had received, an association that was statistically significant (p < 0.001).
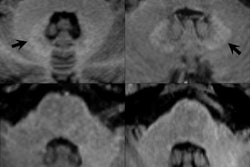
 Unenhanced T1-weighted MR image from a patient who received 37 administrations of a macrocyclic GBCA. Patient has visible dentate nucleus enhancement at last MRI scan (1,359 days after first GBCA-enhanced scan) and no dentate nucleus enhancement at baseline. Image courtesy of Radiology.
Unenhanced T1-weighted MR image from a patient who received 37 administrations of a macrocyclic GBCA. Patient has visible dentate nucleus enhancement at last MRI scan (1,359 days after first GBCA-enhanced scan) and no dentate nucleus enhancement at baseline. Image courtesy of Radiology.The two patients who received 37 and 44 single-dose macrocyclic GBCA injections, respectively, showed visible dentate nucleus enhancement but no enhancement on baseline MR images.
"These results are in agreement with those from the recent study by Stojanov et al, but are in contradiction to results of most published data in which authors concluded that multiple injections of macrocyclic GBCAs do not result in measurable gadolinium retention," the authors wrote. "Our results must be confirmed in larger and better-controlled prospective studies, but they suggest that both linear and macrocyclic GBCAs can cause gadolinium retention, albeit at very different levels of total gadolinium exposure."
Study limitations
Bjørnerud and colleagues cited several limitations of the study, such as the fact that it only included 17 patients with high-grade gliomas.
"Therefore, all patients had known brain neoplasms that could have influenced the time evolution of the MR imaging signal intensity in different parts of the brain," they noted. "For this reason, we did not include measurements in the commonly investigated globus pallidus, because high-grade gliomas frequently affect this region of the brain."
In addition, the two radiologists who visually evaluated the first and last-scan precontrast T1-weighted MR images were not blinded to the order of the images, although they were blinded to the dose given to each patient.
"This could have introduced a bias toward observing less enhancement in the baseline examinations, but it was unlikely to have influenced the relative enhancement scoring of the last-time-point images among patients," the authors wrote.