Provisio Medical's Sonic Lumen Technology (SLT) Intravascular Ultrasound (IVUS) system has been granted 510(k) clearance by the U.S. Food and Drug Administration (FDA).
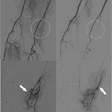
SLT provides automatic, real-time, numeric measurements of the flow lumen of blood vessels without the complexities of image interpretation, the company said. It consists of the SLT IVUS P1 System and the SLT IVUS Support Crossing Catheter; the catheter is an over-the-wire intravascular ultrasound catheter with an ultrasound transducer array at the distal end, which also works as a support crossing catheter. Information from the ultrasound signal is used similarly to sonar technology to measure vessel dimensions in real-time and visualize the peripheral vessel's flow lumen, Provisio Medical noted.