The U.S. Food and Drug Administration (FDA) has warned that Bolton Medical issued an urgent recall of its Relay Pro Thoracic Stent Graft System after three patient deaths were linked to a delivery system failure.
“As of April 23, Bolton Medical has reported three deaths associated with this issue, including one aortic perforation and two conversions to open surgery which resulted in patient deaths due to stroke,” the FDA said, in an April 28 early alert.
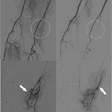
Bolton Medical has reported that in some cases the graft is unable to unclasp from the delivery system because the proximal clasp is disconnected from the outer control tube. The failure cannot be recognized until it occurs mid-procedure and has no device-based bailout solution. Difficulties releasing the stent graft may result in procedural delays, stent graft displacement, or an inability to release the device, potentially requiring conversion to open surgical repair.
The FDA is advising clinicians to consider alternative stent graft options prior to using affected Relay Pro devices until mitigation measures are in place, and to review existing bailout methods in the instructions for use before any procedure.
The FDA’s alert can be found here.