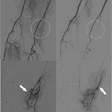
Interventional technology developer Cordis has received an approval recommendation from the Circulatory System Devices Panel of the Food and Drug Administration for its Cordis Carotid System.
The system is intended for use as a minimally invasive treatment alternative to carotid endarterectomy in high-risk patients, according to the Warren, NJ-based subsidiary of Johnson & Johnson.
The product includes two components: the Precise Nitinol Self-Expanding Stent and the Angioguard Emboli Capture Guidewire. These products are currently approved for use in carotid arteries outside the U.S., the company said.
By AuntMinnie.com staff writersApril 22, 2004
Related Reading
Cordis stent gets CE OK, April 8, 2004
Guidant, Johnson & Johnson ink stent deal, February 27, 2004
Cordis stent gets FDA OK, September 5, 2003
Cordis files suit against Boston Scientific, January 15, 2003
Cordis debuts new biliary stent, August 27, 2002
Copyright © 2004 AuntMinnie.com