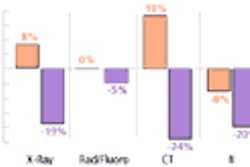
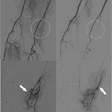
Interventional device developer Boston Scientific of Natick, MA, has received a license for the sale of its Carotid Wallstent Monorail Endoprosthesis from Health Canada’s Therapeutic Products Directorate.
The firm said it received a license for the use of all stent sizes of the Carotid Wallstent system, and that it plans to launch the product immediately in Canada.
By AuntMinnie.com staff writersFebruary 19, 2004
Related Reading
Boston Scientific expands Minnesota operations, February 18, 2004
Boston Scientific posts final 2003 results, February 2, 2004
Boston Scientific gets French reimbursement for Taxus, January 29, 2004
Boston Scientific shows uptick in preliminary 2003 results, January 14, 2004
Boston Scientific, NitroMed extend collaboration, January 8, 2004
Copyright © 2004 AuntMinnie.com