ORLANDO, FL - Computer-aided detection programs originally made their mark in mammography, and are expanding into new applications like chest imaging. Virtual colonoscopy CAD is another promising application that could help radiologists detect more cancers and adenomatous polyps, according to a presentation at this week's Society for Computer Applications in Radiology (SCAR) meeting.
Speaking at a SCAR University educational session, Dr. Ronald M. Summers, Ph.D., of the National Institutes of Health in Bethesda, MD, provided an overview of CAD applications in virtual colonoscopy. While the algorithms underlying the applications are not yet mature, and will require further clinical validation, Summers believes VC CAD has a bright future.
Virtual colonoscopy itself is a relatively new clinical application for detecting colorectal cancer and polyps, but it's beginning to gain traction as a less invasive alternative to optical colonoscopy. The problem is that VC studies can be difficult to read, taking from 10-30 minutes of a radiologist's time to do well. The time requirement can be a drain on a department's resources, and can lead to reader fatigue, Summers said, noting that interreader variability is also an issue.
CAD could potentially make virtual colonoscopy more sensitive and more efficient by enabling radiologists to focus on those areas that a second reviewer -- in this case a computer program -- has highlighted as suspicious. A typical CAD application is designed to detect adenomas -- the type of polyps most likely to develop into adenocarcinoma -- and analyzes them for factors such as surface shape, CT attenuation, and colon wall thickness.
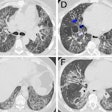
The CAD then highlights suspicious areas using markers on the image, which can then be reviewed in more detail by the radiologist. A 3D display can help the reader make sure that all sections of the colon are being analyzed.
As with other CAD applications, users of VC CAD must negotiate a tricky path between sensitivity and false positives. Published studies of VC CAD's sensitivity range between 65% to 100% for polyps 1 cm and larger, but false positives have ranged from one to seven per patient. In his research, Summers has found that false positives tend to appear in certain specific areas.
"There are common causes of false positives, such as ileocecal valves, rectal tubes, normal folds, stool, and extrinsic compression of the colon by other structures," Summers said. "People are working on ways to get rid of these different false positives. In some cases they're just annoying -- it's easy to recognize the rectal tube -- but in other cases it can be important to distinguish polyps from things like ileocecal valves."
At the National Institutes of Health, Summers and colleagues are working on enhancing VC CAD through developments like polyp segmentation, which attempts to identify the outlines of a polyp to enable measurements of internal polyp characteristics like volume or texture within the polyp. This process can help distinguish true positives from false positives.
Another project is ileocecal valve detection. The valve is visible in nine of 10 patients and accounts for one-fourth of false-positive detections, Summers said. If you segment the valve, you can identify its margins, volume, and CT attenuation, and therefore eliminate some two-thirds of the false positives involving the ileocecal valve. Other false-positive compensation techniques could include rectal tube identification and prone-supine image registration.
Summers provided some advice on clinical users thinking about applying CAD to their practice. He recommends the following scan protocol:
- Thin collimation of 2.5 mm or less
- Slice interval of 1.5 mm or less
- Single breathhold image acquisition
- Low radiation dose, but not so low as to introduce too much noise into the image, which can impede the CAD algorithm
- A cathartic bowel prep, and an oral contrast for fluid and stool tagging (although Summers said he is in the minority on the use of oral contrast)
- No IV contrast
How can CAD be used in a clinical setting as a second reader? First you would review the image without CAD, then review it with CAD to complement your first read. Summers believes that if CAD does not identify an abnormality you found on the first read, you shouldn't change your initial call on the image.
"CAD does miss things, and I don't think it's a good idea to say, 'Oh, CAD didn't find it, it must not be real and I'll change my mind,'" Summers said.
CAD has some blind spots, including the following:
- Polyps under fluid and at the air-fluid boundary (This is a particular problem with oral contrast.)
- Small, sessile, or flat polyps
- Polyps touching or on folds
- The ileocecal valve region, as CAD tends to highlight the ileocecal valve frequently (Polyps touching or on the valve can be missed by the radiologist.)
- Rectal polyps touching the rectal tube
A number of issues need to be resolved before VC CAD can achieve widespread use, Summers said. More clinical studies are needed to validate its effectiveness, and VC CAD software must be integrated with PACS networks to make it easier to use. Insurance reimbursement similar to what CAD enjoys in mammography is also necessary to move the technology into the mainstream, he said.
By Brian Casey
AuntMinnie.com staff writer
June 3, 2005
Related Reading
Support vector machines boost accuracy of VC CAD, May 3, 2005
New VC CAD applications show promise as second reader, March 4, 2005
VC CAD system finds polyps in opacified fluid, January 3, 2005
Low-prep VC study finds CAD can be fooled, December 23, 2004
Mass appeal: VC CAD doesn't stop at polyps, October 10, 2003
Copyright © 2005 AuntMinnie.com