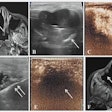
Ultrasound contrast developer Acusphere has signed a collaboration, licensing, and supply agreement with Nycomed for the European development and marketing rights to Acusphere's AI-700 ultrasound contrast agent. AI-700 is currently in phase III clinical trials for assessing myocardial perfusion in the diagnosis of coronary heart disease.
As part of the deal, Nycomed will provide $70 million in licensing fees, research and development funding, and milestone payments of $12 million over the first two years.
Near-term payments to Acusphere include $4 million in upfront license fees and $8 million in R&D funding payable in eight quarterly installments. An additional $58 million in milestone payments are tied to regulatory approvals and achievement of certain sales goals, according to Acusphere of Watertown, MA.
In addition, Acusphere will be paid to manufacture AI-700 for Nycomed, and will receive royalties on Nycomed's sales of the agent. Nycomed will also handle sales, marketing, and the regulatory submissions required for marketing throughout its sales territory, which includes the member states of the European Union, as well as Russia/CIS, and Turkey, Acusphere said.
By AuntMinnie.com staff writersJuly 7, 2004
Related Reading
Ultrasound firm Acusphere launches IPO, October 15, 2003
Acusphere gets financing for development, April 21, 2003
Acusphere launches phase III trials, February 26, 2003
Acusphere raises $15 million, July 24, 2002
Acusphere US contrast agent research presented at ASE, July 19, 2001
Copyright © 2003 AuntMinnie.com