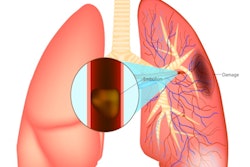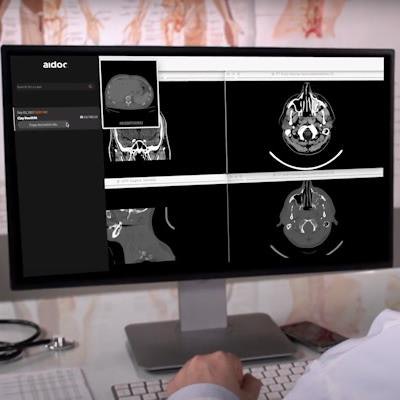
Artificial intelligence (AI) software developer Aidoc has garnered U.S. Food and Drug Administration (FDA) clearance for its computer-assisted triage technology in flagging and notification of incidental pulmonary embolism (PE) on CT scans.
The clearance is the sixth for the company. The software will be available alongside Aidoc's other triage and notification AI algorithms for intracranial hemorrhage, C-spine fractures, large vessel occlusion (LVO), intra-abdominal free gas, as well as PE on dedicated exams, according to the vendor.
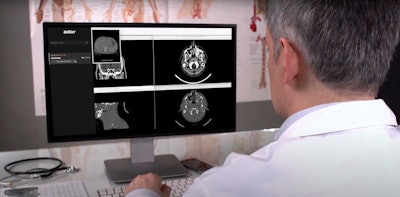 Aidoc's new AI algorithm provides flagging and notification of incidental pulmonary embolism found on CT scans. Image courtesy of Aidoc.
Aidoc's new AI algorithm provides flagging and notification of incidental pulmonary embolism found on CT scans. Image courtesy of Aidoc.Aidoc also noted that last month's decision by the U.S. Centers for Medicare and Medicaid Services to provide new technology add-on payment (NTAP) status for software to detect stroke on CT scans also applies to its AI algorithm for detecting and triaging LVOs. In addition, the firm said it's working to submit other AI technology for possible inclusion in the NTAP reimbursement program.