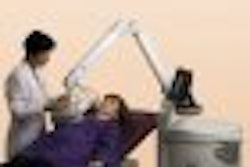
Ultrasound technology developer Misonix reported that it has signed its first deal to provide high-intensity focused ultrasound (HIFU) technology in Hungary.
The SOTE Urological Clinic at the University Hospital of Budapest has begun using the Farmingdale, NY-based company's Sonablate 500 HIFU system to treat prostate cancer, according to Misonix.
By AuntMinnie.com staff writers
November 16, 2006
Related Reading
Misonix posts Q1 sales uptick, November 14, 2006
Misonix begins Hungarian sales, August 4, 2006
Misonix enters French market, July 25, 2006
Misonix takes controlling stake in U.K. distributor, April 11, 2006
Misonix promotes Constantine, November 23, 2005
Copyright © 2006 AuntMinnie.com