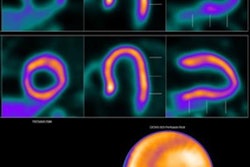
Lantheus Holdings subsidiary Lantheus Medical Imaging has signed a definitive agreement with GE Healthcare to support the worldwide development and commercialization of its flurpiridaz F-18 investigational PET radiopharmaceutical. The firms are also continuing their current supply agreement for other nuclear medicine products.
Following on a term sheet the companies signed in February, the definitive agreement provides for GE to lead and fund the development program of flurpiridaz F-18, including a second phase III clinical study. In addition, GE will have exclusive worldwide rights to commercialize the radiopharmaceutical, a myocardial perfusion imaging (MPI) agent designed to improve the diagnosis of coronary artery disease.
Lantheus will collaborate in both the development and commercialization process through a joint steering committee, and the company also maintains the option to copromote the agent in the U.S. In return, Lantheus receives a $5 million cash payment and up to $60 million in regulatory and sales milestone payments. It will also receive double-digit royalties on U.S. sales and single-digit royalties on sales outside of the U.S, according to the companies.
In a separate development, Lantheus and GE have also extended and expanded their current commercial supply agreement for other nuclear medicine offerings. As a result, Lantheus will continue to supply GE with its TechneLite, gallium-67, and xenon-133 radiopharmaceuticals through December 31, 2020.