MRI radiomics and machine learning can enable identification of inflammatory bowel disease (IBD) subtypes. A study on the research was presented at this week's International Society for Magnetic Resonance in Medicine (ISMRM) meeting in London.
Researchers led by Jun Wang of Lanzhou University Second Hospital in Lanzhou, China, developed machine-learning models based on radiomics analysis of contrast-enhanced MRI of the small bowel. In testing, the best-performing algorithm yielded an area under the curve (AUC) of 0.874 for differentiating ulcerative colitis and Crohn's disease.
"Radiomics may be a potential source of biomarker-based disease classification and diagnosis," Wang said.
The ISMRM is holding this week's meeting in conjunction with the European Society for Magnetic Resonance in Medicine and Biology and the International Society for MR Radiographers and Technologists.
Increasing global burden
The burden of inflammatory bowel disease is increasing globally, with substantially rising levels across many countries and regions, according to Wang. There is currently no single gold-standard diagnostic test for IBD, and there are many disease processes that can be diagnosed as IBD, he said.
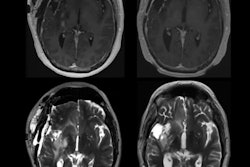
Contrast-enhanced MRI provides better visualization of the entire bowel wall in comparison to conventional T2-weighted imaging. The researchers sought to apply radiomics to the task of distinguishing between ulcerative colitis and Crohn's disease on these MRI exams.
They retrospectively gathered data from 216 IBD patients who had received colonoscopy and contrast-enhanced MRI between 2012 and 2021 at their institution. Of these, 116 had ulcerative colitis and 100 were diagnosed with Crohn's disease.
The contrast-enhanced T1-weighted small-bowel MRI exams were acquired on either an Ingenia CX 3-tesla scanner (Philips Healthcare) or a 1.5-tesla scanner from Siemens Healthineers using a protocol of 1,100 ms repetition time, 374 x 132-mm2 field of view, 80-ms time echo, 105 axial slices, 1.1-mm slice thickness, and a 90-degree flip angle. All patients received 0.1 mmol/kg of gadopentetate dimeglumine (MultiHance, Bracco Imaging).
Next, the researchers used the PyRadiomics software to extract 1,037 radiomics features from manually drawn regions of interest on the images that depicted abnormally thickened intestinal walls. The radiomics features included first-order, texture, and shape features. A least absolute shrinkage and selection operator (LASSO) regression model was then employed to select the 15 best radiomics features and build a radiomics "signature."
This data was used to train six different types of machine-learning models, including logistic regression, K-nearest neighbor, naive Bayes, support vector machine, gradient boosting trees, and decision-tree algorithms. The algorithms were assessed via 10-fold cross-validation.
The researchers found that the logistic-regression algorithm yielded the highest performance on the test set.
| Performance of MRI radiomics machine-learning model in distinguishing IBD subtypes | |
| AUC | 0.874 |
| Sensitivity | 81% |
| Specificity | 78% |
| Accuracy | 80% |
"The radiomics model centered on contrast-enhanced MR showed good performance in differential diagnosis of IBD," he said.
Delving further into the results, the researchers found that nine of the 15 significant radiomics predictors were calculated from decomposed MRI data after the wavelet transform. Junior radiologists often have difficulty in interpreting this data, according to Wang.
"The results indicate that wavelet image decomposition enhanced the discriminatory power of radiomics, possibly due to the noise suppression and identification of frequency bands that better capture the actual imaging phenotype variability," he said. "Enhanced images can reflect intestinal inflammation's more valuable information on blood supply, internal lesions, and more."