NEW YORK CITY - A deep-learning algorithm that analyzes B-mode morphological ultrasound data and patient demographic information can classify the severity of liver fibrosis in cases of chronic liver disease, according to research presented at the American Institute of Ultrasound in Medicine (AIUM) conference.
A team of researchers from Greece trained a deep neural network that could determine if patients were in the early stages of fibrosis or if they had advanced to significant fibrosis. In testing, the algorithm yielded a higher level of performance than existing methods previously reported in the literature, according to presenter Athanasios Angelakis, PhD, from the National Technical University of Athens.
A binary classification problem
Early-stage fibrosis can be reversed with treatment and prevented from reaching cirrhosis. It's also important to be able to diagnose significant fibrosis, classified as Metavir score F0 or F1, from more severe fibrosis (≥ F2).
 Athanasios Angelakis, PhD, from the National Technical University of Athens.
Athanasios Angelakis, PhD, from the National Technical University of Athens."Given an individual's biomarkers, we would like to conclude if his or her fibrosis stage is either [Metavir score] ≤ F1 or ≥ F2," he said in a March 24 presentation "It's a significant binary classification problem."
Traditional methods used for this assessment include biopsy; biochemical tests; imaging, such as shear-wave elastography, strain elastography, and FibroScan transient elastography cutoff values; as well as image analysis or radiomic approaches. Recently, machine learning has also been applied in image analysis and radiomics. However, none of the biochemical, imaging, image analysis/radiomics, or machine-learning methods reported in the literature has yielded an area under the curve (AUC) from receiver operating characteristic analysis of more than 0.87, according to Angelakis.
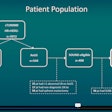
The researchers sought to apply deep learning to this classification task. They used a dataset of 103 patients with chronic liver disease and biopsy-validated fibrosis stages; 62 patients had Metavir scores of F0 or F1, while 41 had scores of at least F2. The deep-learning network was given one demographic parameter (patient gender) and four morphologic parameters -- the longitudinal diameter of the left lobe, caudate lobe, and right lobe of the liver, as well as the spleen. The algorithm then provides its assessment of the patient's fibrosis class (≤ F1 or ≥ F2).
This information could be of particular benefit to inexperienced users, according to the researchers.
"It reduces the experience [needed] for the examiner [to perform these studies]," Angelakis said. "One of the goals of using deep learning in ultrasound in general and shear-wave elastography is to reduce this need for experience with the exam."
After performing tenfold cross-validation, the researchers found that the algorithm yielded a high level of accuracy for liver fibrosis classification:
- Sensitivity: 90.2%
- Specificity: 91.9%
- Positive predictive value: 88.1%
- Negative predictive value: 93.4%
- AUC: 0.9126
They acknowledged, though, that more patients are needed to further validate the algorithm.
The future
The ultimate holy grail for a deep-learning algorithm in the liver would be to incorporate patient demographic data such as gender and age, along with morphologic, hemodynamic, biochemical, image analysis, and elastographic data. Such an algorithm, which would have many layers, would not only classify fibrosis but also steatosis, portal hypertension, and inflammation, Angelakis said.
"We have already created algorithms and models that also attack the steatosis problem, because our datasets are bigger now," he said.
Deep learning and data science will likely take on a significant role in ultrasound in the future, according to Angelakis.
"The output from the [deep-learning] model will be one more input for the expert [to consider during interpretation]," he said. "That is how we reduce the expertise [requirement] for the reader. Looking ahead 10 years from now, maybe less, data science will also be used during the manufacturing of ultrasound devices and it will be in every [aspect of the ultrasound acquisition and interpretation process]."