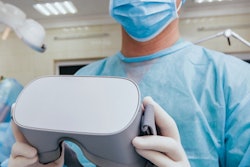
Canadian virtual reality software developer Luxsonic said that its SieVRt virtual reality (VR) radiology software platform has received Health Canada approval as a class 2 medical device for use in diagnostic radiology.
The software suite, which can be operated using a virtual-reality headset such as Oculus, provides advanced medical imaging visualization tools for students, trainees, and physicians, according to the Saskatoon, Saskatchewan-based company. The newly cleared Diagnostic module enables radiologists to bring their own personalized virtual office with them wherever they go and remotely diagnose patients, Luxsonic said.
Other currently available SieVRT modules in North America include Basic, Education, and Collaboration. Luxsonic said it hopes to receive U.S. Food and Drug Administration clearance for SieVRt in early 2022. Massachusetts General Hospital, Cleveland Clinic, and McMaster University are all currently trialing the software, according to the firm.