Researchers from Pennsylvania have developed a way to create realistic 3D-bioprinted heart models that reproduce not only the cardiac texture and microstructure of MR images but also cardiac function and cell growth. Their new 3D bioprinting technique is the subject of an article published in the August 2 issue of Science.
The group from Carnegie Mellon University in Pittsburgh previously developed a method for creating a type of bioink -- i.e., collagen -- that replicated the structural environment of the heart and supported the growth and differentiation of living cells. The technique, called freeform reversible embedding of suspended hydrogels (FRESH v1.0), relied on rapid pH changes to drive the production of collagen, which researchers then used as the building block for 3D-bioprinted hearts.
Collagen is the heart's main extracellular component and is ideal for use in 3D bioprinting because it provides sound structural organization of cell and tissue compartments. Collagen can also serve as a depot for live cells, but generating complex 3D-bioprinted models with this soft material has proved challenging, noted corresponding author Adam Feinberg, PhD, and colleagues.
In the current study, Feinberg and colleagues adjusted their FRESH v1.0 technique in such a way that it could generate collagen filaments with greater uniformity and higher resolution to produce models that would more accurately reflect the properties of a real human heart.
The latest iteration of their technique (FRESH v2.0) allowed them to 3D bioprint parts of the human heart at a resolution of 20 µm -- a considerable improvement from the 100- to 500-µm resolution achieved with traditional extrusion-based 3D printing and the 250-µm resolution of models made using FRESH v1.0 (Science, August 2, 2019, Vol. 366:6452, pp. 482-487).
"This platform technology actually allows us to start ... fabricating these tissues that are complex in their material composition as well as their architecture, and this allows us to create three-dimensional organ functions that more closely mimic what is typically found in the human body," first author Andrew Lee, a doctoral candidate at the university, said in a statement.
Indeed, the researchers were able to use the technique to create a 3D-bioprinted mouse heart model that consisted of a combination of collagen bioink, proangiogenic molecules, and live heart cells. Quantitative analysis revealed significant cell growth in the model after three and seven days, compared with the control model (p < 0.0001), and widespread vascularization with red blood cells visible in the vasculature after 10 days in vivo.
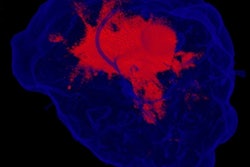
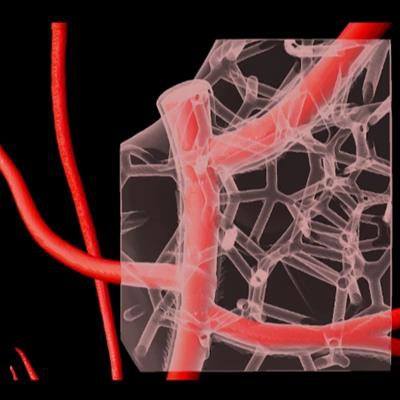
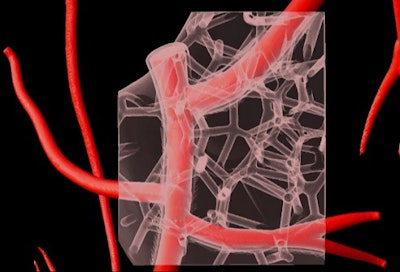 Virtual depiction of part of a 3D-bioprinted model of cardiac vasculature made with collagen bioink. Image courtesy of Carnegie Mellon University College of Engineering.
Virtual depiction of part of a 3D-bioprinted model of cardiac vasculature made with collagen bioink. Image courtesy of Carnegie Mellon University College of Engineering.The group subsequently 3D bioprinted a model of the left ventricle of a human heart using a combination of collagen bioink and actual heart muscle cells derived from stem cells. The left ventricles began to visibly contract just four days after cultivation, and histology showed that the collagen had become synchronous with the heart cells at day seven.
"When printed with relevant cells such as embryonic stem cells, these 3D-printed tissues generated functional outcomes expected of the human heart, such as synchronized beating and the opening and closing of printed valves during pulsatile flow," Queeny Dasgupta, PhD, and Lauren Black III, PhD, wrote in an invited commentary.
Micro-CT scans of yet another 3D-bioprinted model -- this time, a full neonatal human heart -- confirmed that measurements of the model were almost identical to those from virtual 3D models of the original cardiac MR images.
"We now have a foundation and the ability to print functional parts of the heart so that we can think about starting to put those together to move beyond just tissues and start to achieve this reality of creating functional organs," Feinberg said. "While we've not yet achieved printing a whole adult-sized functional heart, what we've done is taken critical steps along that path."