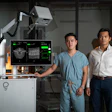
RIS/PACS vendor Novarad has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its OpenSight augmented reality software.
OpenSight is designed to improve surgical procedures by enhancing accuracy and shortening operation times, according to the company. It works with Microsoft's HoloLens virtual reality headset to enable clinicians to view 2D, 3D, and 4D medical images that are superimposed on real-world views of patients, in real-time. The combination of actual and virtual reality avoids some of the disorientation that can occur in purely virtual environments.
Novarad believes that OpenSight enables physicians to better understand anatomic relationships because images are co-localized to the patient. Preoperative planning can be performed with OpenSight, highlighting relevant anatomy and critical structures that surgeons should avoid. The software also includes positioning virtual tools and guidance systems to plan surgical approaches more accurately.
Multiple HoloLens headsets running OpenSight can be shared between users, which can improve the training of less-experienced residents. Novarad also offers a teaching version of OpenSight that allows medical students to perform virtual dissections on cadavers.