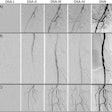
The European Union Clinical Trial Directive and the GCP Directive are having major implications on how clinical trials are being managed in the EU. Several additional CT/GCP Directive guidelines have recently been released to complete the directives' requirements. EU countries have now transposed the main requirements of the CT directive into national legislation, but in many countries further degrees, guidelines and other documents are being issued to complete the implementation. This transition period (until all the national requirements are fully implemented) has proven a challenge for many organizations seeking to carry out clinical trials in the EU. The Timing of the workshop is ideal in that it provides an excellent opportunity to share experiences and discuss the current issues.
EU Clinical Trial & GCP Directives - An Interactive Workshop
May 9th, 2007May 10th, 2007
London, --
GB
Latest in Home