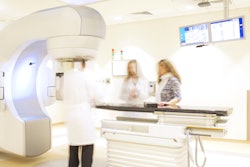
Halcyon radiotherapy system
Siemens Healthineers
Varian, a Siemens Healthineers company, has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for enhancements to its Halcyon radiotherapy system.
The clearance includes upgrades to the system's PerfectKinetix Dynamic Couch and its Identify motion‑management capabilities. Combined with HyperSight conebeam CT imaging, the upgrades support treatment of both routine and complex cancer cases.
Halcyon consumes up to 70% less power and requires 65% less concrete than a C‑arm linac, while its reduced weight lowers freight‑related emissions, according to the firm.