Radiation therapy firm TomoTherapy said it has received U.S. Food and Drug Administration (FDA) clearance for TomoDirect, a new mode for its Hi-Art radiation therapy system.
The Madison, WI, company said that TomoDirect is a discrete-angle, sliding-beam delivery mode for Hi-Art that complements the system's existing helical TomoTherapy mode. TomoDirect allows clinicians to choose several discrete angles, as well as the optimal modulation level required for delivery, the company said. It's expected to provide significant time savings in both the planning and delivery phases for several clinical scenarios, including whole-breast irradiation and palliative treatments.
TomoDirect utilizes the same binary multileaf collimator and CT-style gantry technology as helical TomoTherapy, and both modes share a consistent treatment planning and delivery process. The company said that Hi-Art's treatment modes are also being expanded to include 3D conformal delivery.
Related Reading
TomoTherapy to shuffle management, August 26, 2008
TomoTherapy posts Q2 results, August 1, 2008
TomoTherapy readies TQA, July 25, 2008
TomoTherapy to buy Japanese distributor, July 9, 2008
TomoTherapy posts Q1 prelims, buys Chinese linac firm, April 18, 2008
Copyright © 2008 AuntMinnie.com





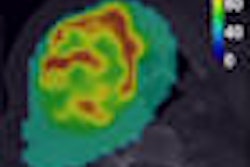
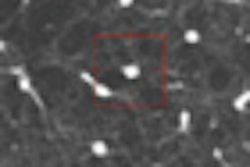
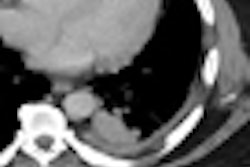





![A 53-year-old patient (patient number four) with a recurrent pituitary adenoma with extension of a cystic component of disease to the medial temporal lobe apparent on MRI (contoured in blue), and extension of disease to the left sphenoid bone and orbital apex apparent on [68Ga]Ga-DOTA-TATE (contoured in yellow).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/04/pituitary-tumor.QGsEnyB4bU.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)





