Swedish radiation oncology firm Elekta has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its Volumetric Intensity Modulated Arc Therapy (VMAT) technology.
VMAT will be available via the company's Synergy digital linear accelerator offering, according to the Stockholm-based firm.
Related Reading
Elekta launches virtual clinic, June 13, 2008
Elekta wins Louisiana radiotherapy contract, June 5, 2008
Elekta receives Australian order, May 2, 2008
Elekta readies first order for New Zealand, April 25, 2008
Elekta completes CMS purchase, March 5, 2008
Copyright © 2008 AuntMinnie.com
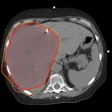
![A 53-year-old patient (patient number four) with a recurrent pituitary adenoma with extension of a cystic component of disease to the medial temporal lobe apparent on MRI (contoured in blue), and extension of disease to the left sphenoid bone and orbital apex apparent on [68Ga]Ga-DOTA-TATE (contoured in yellow).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/04/pituitary-tumor.QGsEnyB4bU.jpg?auto=format%2Ccompress&fit=crop&h=100&q=70&w=100)







![A 53-year-old patient (patient number four) with a recurrent pituitary adenoma with extension of a cystic component of disease to the medial temporal lobe apparent on MRI (contoured in blue), and extension of disease to the left sphenoid bone and orbital apex apparent on [68Ga]Ga-DOTA-TATE (contoured in yellow).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/04/pituitary-tumor.QGsEnyB4bU.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)