Radiation therapy vendor Varian Medical Systems has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its RapidArc radiotherapy technology.
The technology is designed to deliver image-guided intensity-modulated radiation therapy (IMRT) two to eight times faster than conventional IMRT or helical tomotherapy, according to the Palo Alto, CA-based vendor.
Related Reading
Varian buys Chinese distributor, December 26, 2007
Kyron inks deal with Varian, December 24, 2007
Varian gets FDA OK for scanning algorithm, December 14, 2007
Varian receives Swiss orders, December 12, 2007
Varian scores U.K. install, November 29, 2007
Copyright © 2008 AuntMinnie.com







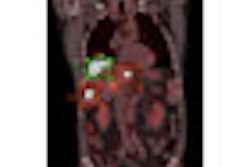





![A 53-year-old patient (patient number four) with a recurrent pituitary adenoma with extension of a cystic component of disease to the medial temporal lobe apparent on MRI (contoured in blue), and extension of disease to the left sphenoid bone and orbital apex apparent on [68Ga]Ga-DOTA-TATE (contoured in yellow).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/04/pituitary-tumor.QGsEnyB4bU.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)




