Radiation therapy firm Varian Medical Systems of Palo Alto, CA, has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for a new proton scanning dose algorithm.
The algorithm is designed to give clinicians greater control over how proton beams are used to target tumors and other abnormalities. It is incorporated into Varian's Eclipse treatment planning software, which helps caregivers plan for intensity-modulated proton therapy by optimizing dose distributions in three dimensions to protect surrounding healthy tissues.
The proton scanning technique is intended to enhance current proton delivery methods that require a therapist to enter and exit the treatment room numerous times throughout a treatment to adjust physical compensators for processing proton beams.
Related Reading
Varian receives Swiss orders, December 12, 2007
Varian scores U.K. install, November 29, 2007
Varian adds PaxScan models, November 22, 2007
Varian inks long-term U.K. pact, October 30, 2007
Varian to launch new IMRT at ASTRO, October 25, 2007
Copyright © 2007 AuntMinnie.com


![A 53-year-old patient (patient number four) with a recurrent pituitary adenoma with extension of a cystic component of disease to the medial temporal lobe apparent on MRI (contoured in blue), and extension of disease to the left sphenoid bone and orbital apex apparent on [68Ga]Ga-DOTA-TATE (contoured in yellow).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/04/pituitary-tumor.QGsEnyB4bU.jpg?auto=format%2Ccompress&fit=crop&h=100&q=70&w=100)



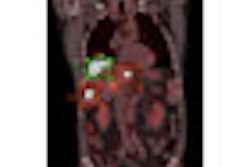



![A 53-year-old patient (patient number four) with a recurrent pituitary adenoma with extension of a cystic component of disease to the medial temporal lobe apparent on MRI (contoured in blue), and extension of disease to the left sphenoid bone and orbital apex apparent on [68Ga]Ga-DOTA-TATE (contoured in yellow).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/04/pituitary-tumor.QGsEnyB4bU.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)