Radiation-therapy developer Proxima Therapeutics of Alpharetta, GA, said it has received Food and Drug Administration marketing clearance for elliptical-shaped balloon catheters for its MammoSite catheter-based radiation treatment device.
The new elliptical balloon catheters will enable more women to be eligible for partial breast irradiation following lumpectomy. The older spherical MammoSite balloon catheters, which have been used to treat thousands of patients, will still be available, the company said.
By AuntMinnie.com staff writersApril 1, 2004
Related Reading
MammoSite RTS treats first Canadian patient, September 17, 2003
Proxima is cleared for MammoSite RTS, May 7, 2002
Copyright © 2004 AuntMinnie.com





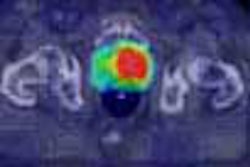
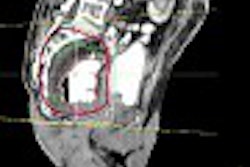





![A 53-year-old patient (patient number four) with a recurrent pituitary adenoma with extension of a cystic component of disease to the medial temporal lobe apparent on MRI (contoured in blue), and extension of disease to the left sphenoid bone and orbital apex apparent on [68Ga]Ga-DOTA-TATE (contoured in yellow).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/04/pituitary-tumor.QGsEnyB4bU.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)