MR proton density fat fraction (PDFF) imaging helps predict liver health, researchers have found.
The MRI PDFF measure could improve patient care by giving clinicians a more accurate picture of liver disease, a team led by Tianyi Xia, MD, of Zhongda Hospital School of Medicine, Southeast University, in Nanjing, China. The study results were published October 24 in Radiology.
"We found … that liver MRI PDFF was a causal mediator between potential risk factors and several liver disease risks," the group wrote.
Hepatic steatosis is one of the anatomic characteristics of nonalcoholic fatty liver disease, and it is the "leading cause of chronic liver disease globally," the authors explained. There are a variety of ways to assess liver fat content, but MRI PDFF has shown high diagnostic precision.
"Liver MRI PDFF provides valuable information regarding triglyceride accumulation within hepatic tissue," the authors wrote. "Importantly, reductions in MRI PDFF have shown a strong correlation with improvements in hepatic steatosis and other histologic features like fibrosis, ballooning degeneration, and lobular inflammation."
Research on the relationship between liver MRI PDFF and liver disease is lacking, according to Xia and colleagues. The team conducted a study to investigate any connection between the two, using data from U.K. Biobank (a U.K. biomedical database that consists of genetic and health information from 500,000 participants) and FinnGen (a Finnish biobank, also with 500,000 participants). Summary-level data included information from 32,858 patients and individual-level data from 378,436 patients.
The investigators found that genetically predicted high liver MRI PDFF was associated with increased risk of the following diseases:
| Odds of increased risk of disease in patients with high liver MRI PDFF | ||
|---|---|---|
| Disease |
Odds ratio |
p-value |
| Nonalcoholic steatohepatitis |
7.7 |
< 0.001 |
| Malignant liver neoplasm |
4.5 |
< 0.001 |
| Nonalcoholic fatty liver disease |
4.4 |
< 0.001 |
| Cirrhosis of the liver |
3.8 |
< 0.001 |
| Fibrosis of the liver |
3.6 |
0.002 |
| Fibrosis and cirrhosis of the liver |
3 |
< 0.004 |
| Alcoholic liver disease |
1.9 |
< 0.001 |
They also reported data that, at liver MRI PDFF on the individual participant level, showed genetically predicted high-density lipoprotein cholesterol, type 2 diabetes, and waist-to-hip ratio were linked to fibrosis and cirrhosis of the liver, cirrhosis of the liver, and nonalcoholic fatty liver disease (all p < 0.05).
"Our study provides evidence that MRI PDFF has the potential to improve liver imaging-level prediction and interventions for liver health, which may have important implications in routine clinical practice," the group concluded.
The complete study can be found here.



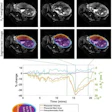
![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=100&q=70&w=100)



![Overview of the study design. (A) The fully automated deep learning framework was developed to estimate body composition (BC) (defined as subcutaneous adipose tissue [SAT] in liters; visceral adipose tissue [VAT] in liters; skeletal muscle [SM] in liters; SM fat fraction [SMFF] as a percentage; and intramuscular adipose tissue [IMAT] in deciliters) from MRI. The fully automated framework comprised one model (model 1) to quantify different BC measures (SAT, VAT, SM, SMFF, and IMAT) as three-dimensional (3D) measures from whole-body MRI scans. The second model (model 2) was trained to identify standardized anatomic landmarks along the craniocaudal body axis (z coordinate field), which allowed for subdividing the whole-body measures into different subregions typically examined on clinical routine MRI scans (chest, abdomen, and pelvis). (B) BC was quantified from whole-body MRI in over 66,000 individuals from two large population-based cohort studies, the UK Biobank (UKB) (36,317 individuals) and the German National Cohort (NAKO) (30,291 individuals). Bar graphs show age distribution by sex and cohort. BMI = body mass index. (C) After the performance assessment of the fully automated framework, the change in BC measures, distributions, and profiles across age decades were investigated. Age-, sex-, and height-adjusted body composition reference curves were calculated and made publicly available in a web-based z-score calculator (https://circ-ml.github.io).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/05/body-comp.XgAjTfPj1W.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)