New research guidelines for classifying whether individuals have Alzheimer's disease rely heavily on the use of biomarkers from imaging scans. The new framework was published in the April issue of Alzheimer's & Dementia: The Journal of the Alzheimer's Association.
The guidelines for the research community were published jointly by the U.S. National Institute on Aging (NIA) and the Alzheimer's Association (AA). They use the presence or absence of beta amyloid, tau protein, and neurodegeneration, along with analysis of cerebrospinal fluid samples, to determine whether a person has Alzheimer's disease and, if so, the severity of the condition (Alzheimers Dement, Vol. 14:4, pp. 535-562).
From imaging measurements, the researchers created eight sample patient profiles. For example, if a person had a positive beta-amyloid biomarker but no tau protein, he or she would be categorized as having "Alzheimer's pathologic change." People with both beta-amyloid and tau biomarkers would be considered to have Alzheimer's disease.
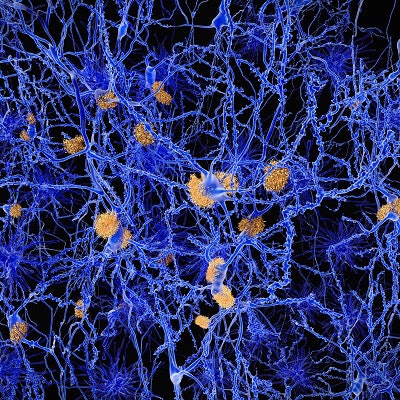
The accumulation of beta amyloid and tau protein in the brain is associated with the onset of Alzheimer's and cognitive impairment. Neurodegeneration or neuronal injury, such as head trauma, is linked to the condition but to a lesser degree.
"In the context of continuing evolution of Alzheimer's research and technologies, the proposed research framework is a logical next step to help the scientific community advance in the fight against Alzheimer's," said Dr. Richard Hodes, director of the NIA, in a release from the institute. "The more accurately we can characterize the specific disease process pathologically defined as Alzheimer's disease, the better our chances of intervening at any point in this continuum, from preventing Alzheimer's to delaying progression."
Through the new framework, the researchers hope to better understand the process that leads to cognitive impairment and dementia. In turn, the information could help improve treatment to slow or halt the progression of the disease.
NIA and AA researchers noted that this approach is for research purposes only and should not yet be considered for the accurate diagnosis of Alzheimer's. Further testing and validation are needed before the guidelines could be confidently implemented in the clinical setting, they added.