An experimental PET radiotracer can predict whether patients will respond early to new immunotherapy treatments for metastatic skin cancer, according to a study published September 9 in the Journal of Nuclear Medicine.
In a pilot study, Dutch researchers performed PET scans with an experimental tracer called F-18 BMS986192 before and after patients received treatment with immune checkpoint inhibitors (ICIs) for metastatic melanoma, or skin cancer that has spread to other locations.
They found the technique predicted whether patients were responding to treatment in weeks rather than months using standard imaging approaches such as CT or MRI.
"Baseline F-18 BMS986192 uptake was able to predict an ICI treatment-induced reduction in lesion volume," wrote first author Pieter Nienhuis of University Medical Center Groningen, Netherlands, and colleagues.
Immune checkpoint inhibitors are a group of new immunotherapy treatments that work by triggering immune cells called T cells to attack cancer cells. They unblock signals that normally keep the immune system in check. Studies show that up to 60% of patients with metastatic melanoma respond to treatment with immunotherapy.
However, it often takes months before a patient's response to immune checkpoint inhibitors can be detected by CT or MR imaging, and thus fast-growing lesions that require additional treatment may go undetected, according to the authors.
The F-18 BMS986192 tracer was synthesized by Bristol Myers Squibb researchers in 2018 and evaluated in a preclinical study published by German authors earlier this year. In this study, the researchers aimed to test for the first time how the tracer performs in patients.
The researchers enrolled eight patients with confirmed metastatic melanoma. Patients underwent whole-body F-18 BMS986192 scans with a PET/CT scanner (Biograph mCT64 or mCT40, Siemens Healthineers) before and six weeks after starting ICI therapy.
Patients received immunotherapy either with nivolumab (brand name Opdivo) or a combination of nivolumab and ipilimumab (brand name Yervoy), two immunotherapy treatments recently approved by the U.S. Food and Drug Administration.
The highest uptake of the tracer was seen in the spleen, bone marrow, kidneys, and liver. Tracer uptake in tumor lesions was heterogeneous. In total, 42 tumor lesions were identified in patients at baseline, with most lesions in the lungs (n = 21) and brain (n = 14). Tracer uptake was similar between tumor locations.
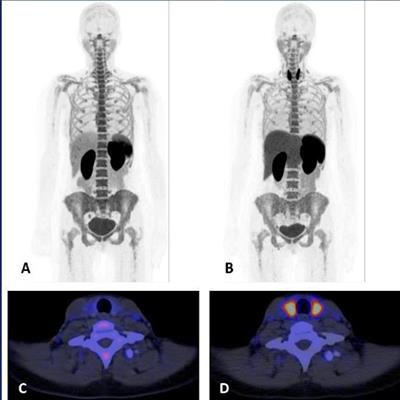
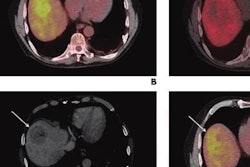
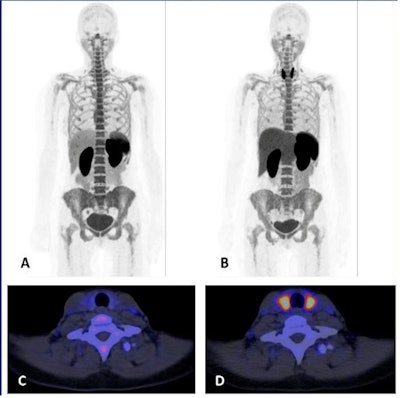 Images A-D show F-18 BMS986192 scans of a patient who developed ICI-related thyroiditis with increased uptake on treatment at six weeks (B,D) compared to baseline (A,C). Image courtesy of the Journal of Nuclear Medicine.
Images A-D show F-18 BMS986192 scans of a patient who developed ICI-related thyroiditis with increased uptake on treatment at six weeks (B,D) compared to baseline (A,C). Image courtesy of the Journal of Nuclear Medicine.The researchers found F-18 BMS986192 uptake in lesions at baseline was negatively correlated with the change in the size of tumors at six weeks. Receiver operating characteristic (ROC) analysis demonstrated that F-18 BMS986192 uptake can discriminate between responding and nonresponding lesions with an area under the curve of 0.82.
"We found heterogeneous uptake of F-18 BMS986192 in metastatic lesions at all locations, which is in line with a heterogeneous response to immune checkpoint inhibitor therapy that is often observed in melanoma patients," the researchers wrote.
Importantly, on follow-up F-18 BMS986192 PET scans of two patients, immune checkpoint inhibitor-related toxicity (thyroiditis and colitis) was detected, the authors added.
This pilot study was the first to investigate F-18 BMS986192 PET imaging before and during immune checkpoint inhibitor therapy in patients with metastatic melanoma, including patients with brain metastases, the authors stated.
"The preliminary results of this pilot study warrant further evaluation of F-18 BMS986192 PET as a noninvasive imaging tool," the researchers concluded.

















![A 53-year-old patient (patient number four) with a recurrent pituitary adenoma with extension of a cystic component of disease to the medial temporal lobe apparent on MRI (contoured in blue), and extension of disease to the left sphenoid bone and orbital apex apparent on [68Ga]Ga-DOTA-TATE (contoured in yellow).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/04/pituitary-tumor.QGsEnyB4bU.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)