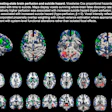
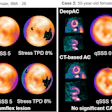
The Society of Nuclear Medicine and Molecular Imaging (SNMMI) is teaming up with Zionexa to urge the U.S. Centers for Medicare and Medicaid Services (CMS) to properly reimburse Cerianna, a radiolabeled F-18 fluoroestradiol radiotracer indicated for use with PET.
The imaging agent is used to detect estrogen receptor-positive lesions as an adjunct to biopsy in patients with recurrent or metastatic breast cancer. At the moment, the current rate of reimbursement is $0.752 per millicurie for Medicare patients instead of $608.33 per millicurie.
CMS said any corrections would be retroactive to January 1, but the correction isn't likely to happen until March for April or July implementation, according to the SNMMI.