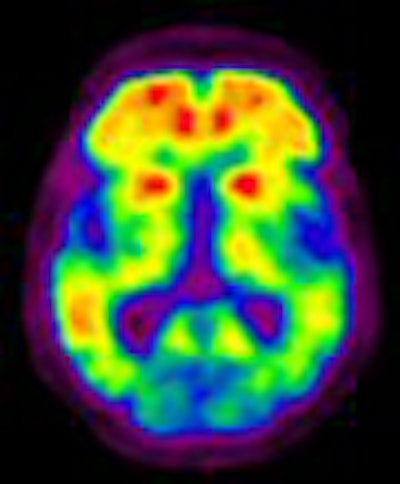
Two studies by Australian researchers are shedding light on the cause of Alzheimer's disease and the potential of a new biomarker to detect the debilitating condition, which may be prevalent as many as 10 years before symptom onset.
Alzheimer's disease is a rapidly increasing healthcare concern around the world. In the U.S., an estimated 5 million people currently are afflicted with Alzheimer's disease. Figures from the Alzheimer's Association also project as many as 16 million people could have Alzheimer's in the U.S. alone by the year 2050.
That forecast strongly supports the need for early detection of Alzheimer's, as well as subsequent therapy and treatment to minimize its impact on the elder population and healthcare delivery systems.
PiB and PET
In a study presented at the 2007 SNM meeting in Washington, DC, researchers at Austin Hospital in Melbourne, Victoria, established a connection between the presence of the toxic brain protein beta-amyloid and Alzheimer's. With Alzheimer's, protein amyloid plaques accumulate between nerve cells in the brain. A healthy brain normally would eliminate the beta-amyloid protein, which forms as hard, insoluble plaques and is associated with brain dysfunction.
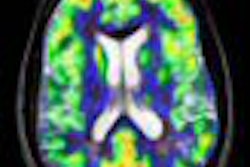
The study used the imaging agent C-11 PiB (Pittsburgh Compound B) and PET imaging to detect early pathological changes of Alzheimer's disease before the development of dementia.
Beta-amyloid is present in 25% of seemingly normal people at age 75 who also show a prevalence of Alzheimer's at age 85. Prior to the development of C-11 PiB and PET, the presence of beta-amyloid only could be confirmed through an autopsy.
"Diagnosis of dementia is too late. To make a diagnosis of Alzheimer's disease, the patient has to be demented," said lead author Dr. Christopher Rowe, director of the nuclear medicine department and Centre for PET at Austin Hospital. "The postmortem data suggest that this process begins for over a decade with the deposition of amyloid seen in postmortem studies in various patients."
The researchers reviewed 34 healthy patients, 44 patients with mild cognitive impairment (MCI), and 44 subjects with mild to moderate Alzheimer's disease. The entire study group underwent cognitive assessment, and beta-amyloid burden was quantified using standard uptake value normalized to cerebellar cortex (SUVR) 40 to 70 minutes postinjection. The mean of frontal, posterior cingulated, parietal, lateral temporal, and occipital regions were used for analysis (see images below).
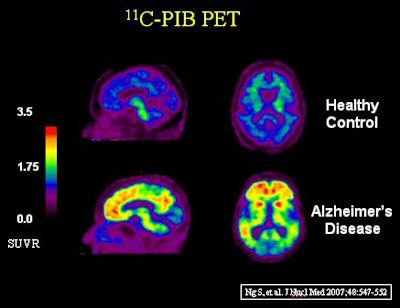 |
| Images courtesy of SNM and Austin Hospital. |
In their analysis, the researchers concluded there is a pathological role for beta-amyloid accumulation in Alzheimer's by showing that relationship in mild cognitive impairment. In addition, the connection was absent in later stages of Alzheimer's disease, as dementia develops.
New F-18 ligand
In related research, Rowe also presented at SNM 2007 the first results from human studies of an F-18 PET ligand for brain beta-amyloid imaging. The molecular imaging agent F-18 AV-1/ZK, currently being developed by Philadelphia-based Avid Radiopharmaceuticals, is designed to target amyloid plaques in the brain.
While C-11 PiB PET imaging has proved useful in detecting beta-amyloid in vivo in Alzheimer's disease, it does have one drawback. The 20-minute half-life of C-11 restricts the use of PiB to centers with an onsite cyclotron.
In the study, five mild Alzheimer's subjects and five healthy control patients underwent PET imaging over three hours after injection of 300 MBq of the F-18 ligand. Distribution volume ratios (DVRs) were calculated through graphical analysis using the cerebellum as input function.
Researchers found that all Alzheimer's subjects showed neocortical binding, with the greatest concentrations in the precuneus and posterior cingulated and frontal cortex. Binding also was prevalent in the lateral temporal and parietal, with relative sparing of the sensorimotor cortex.
The healthy control group showed no binding in cortical or subcortical gray matter, and those scans were distinguishable from the Alzheimer's subjects. Cerebellar cortex showed no retention in either group.
Rowe noted that significantly higher neocortical DVRs were observed in Alzheimer's subjects (1.84 ± 0.20) when compared with the healthy control group (1.3 ± 0.17). Cortical uptake to cerebellar cortex ratio (SUVR) at 90- to 120-minute postinjection gave similar results to DVRs.
'No adverse effects'
The study concluded that the beta-amyloid burden can be quantified in Alzheimer's with AV-1/ZK and the pattern of binding closely matches that of C-11 PiB. "We have observed no adverse effects with this radiopharmaceutical. AV-1/ZK demonstrates a robust increase in retention in Alzheimer's patients with similar appearance to PiB," Rowe said. "No cortical binding was seen in frontal temporal dementia. Therefore, AV-1/ZK appears to be a safe and reliable biomarker for assessing Alzheimer's."
When asked after the SNM presentation what he thought he was seeing in patients who are considered normal but display significant uptake of the compound, Rowe said he thinks "we are seeing the very first stage of Alzheimer's disease pathology."
"It is also consistent with the population studies that show 25% of people over the age of 75 have amyloid infarcts in their brain, which happens to correspond with the incidence of Alzheimer's disease 10 years later. There is a lot circumstantial evidence," he noted.
Research support for the second study came from Neurosciences Victoria, Austin Hospital Medical Research Foundation, and the University of Melbourne.
By Wayne Forrest
AuntMinnie.com staff writer
June 26, 2007
Related Reading
First PET/MRI brain images debut at SNM 2007, June 15, 2007
New tests may help predict Alzheimer's: conference, June 12, 2007
Avid releases results for PET Alzheimer's agent, March 23, 2007
Schering, Avid partner on Alzheimer's agents, July 17, 2006
Copyright © 2007 AuntMinnie.com













![A 53-year-old patient (patient number four) with a recurrent pituitary adenoma with extension of a cystic component of disease to the medial temporal lobe apparent on MRI (contoured in blue), and extension of disease to the left sphenoid bone and orbital apex apparent on [68Ga]Ga-DOTA-TATE (contoured in yellow).](https://img.auntminnie.com/mindful/smg/workspaces/default/uploads/2026/04/pituitary-tumor.QGsEnyB4bU.jpg?auto=format%2Ccompress&fit=crop&h=112&q=70&w=112)