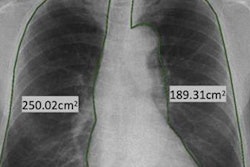
Digital x-ray developer Thales has received clearance from the U.S. Food and Drug Administration (FDA) to market its ArtPix DRF x-ray platform in the U.S.
ArtPix DRF provides dynamic x-ray applications, including high-quality performance and imaging for fluoroscopy, radiography, and angiography. The first system was installed six months ago in Europe, the company noted.
Technologists can use the x-ray system to customize image preferences, user interfaces, and application capabilities. The system also features applications to control the generator and remote tables.
ArtPix DRF will be sold in the U.S. by Thales' original equipment manufacturer partners.