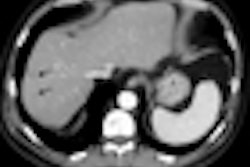
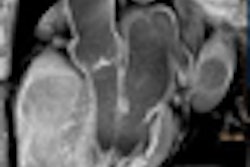
An editorial in this week's New England Journal of Medicine praises the U.S. Centers for Medicare and Medicaid Services' (CMS) recent decision to decline reimbursement for screening virtual colonoscopy (also known as CT colonography or CTC). But a CTC practitioner counters that the agency ignored evidence of the procedure's effectiveness.
According to the May 27 editorial, the "pivotal, overarching concern" at CMS was the lack of CTC trials specifically examining the older Medicare-age population; the average age of the participants in the trials CMS evaluated was 60.1 years, younger than average Medicare recipient, the authors wrote.
Based on the lack of data, editorial authors Dr. Sanket Dhruva, Dr. Steve Phurrough, Dr. Rita Redberg, and Dr. Marcel Salive called the May 12 denial of coverage "a long-overdue step toward meaningful validation of clinical trial evidence in Medicare beneficiaries."
Dhruva is a resident at the University of California, San Francisco (UCSF). Phurrough is a medical officer at the Agency for Healthcare Research and Quality in Rockville, MD.
Redberg is a professor of medicine at UCSF, and Salive is the director of the Division of Medical and Surgical Services at CMS in Baltimore.
CMS' new rigor is a recent phenomenon, the authors noted. "These findings suggest that many previously approved interventions may lack evidence of benefit in the Medicare population -- the group for which U.S. taxpayers are footing the bill."
Before approval is considered for future technologies submitted for coverage, researchers need to "carefully consider the epidemiology of the relevant disease and to ensure that studies are adequately powered to provide meaningful data on discrete subgroups," Dhruva and colleagues wrote.
The authors also suggest that CMS' stricter standards are fragile, and may yet succumb to politics.
"Powerful pressure will inevitably be applied to the CMS. Indeed, after the agency published its draft decision in February, proponents of CT colonography, in a now-familiar pattern, quickly mobilized," the editorial said. "More than 350 comments were submitted to the CMS by interest groups, many with a financial stake in use of the technology."
Citing the agency's retreat from a draft decision limiting coronary CT angiography under similar pressure, they authors stated that "healthcare reform must include explicit authority for the CMS to consider costs in all its coverage decisions in order to assess the true value of a given procedure."
Radiologist responds
"How ironic that the CMS Coverage and Analysis Group is claiming that their decision not to cover screening CTC is based on the evidence -- when that is precisely what they chose to ignore," countered Dr. Perry Pickhardt, associate professor of radiology at the University of Wisconsin in Madison. Pickhardt was the lead author of a recent study in the American Journal of Roentgenology that modeled the cost-effectiveness of CTC in a Medicare-aged population.
"It is also rather curious (if not self-serving) how they 'applaud this landmark decision' in third-person narrative, when two of the authors were actually the ones who made this short-sighted decision," Pickhardt wrote in an e-mail to AuntMinnie.com.
Related Reading
CTC advocates urge CMS to scrap decision, May 14, 2009
CMS rejects Medicare coverage for virtual colonoscopy, May 12, 2009
Pressure builds on CMS to pay for VC, April 2, 2009
CMS rejects case for virtual colonoscopy reimbursement, February 12, 2009
American Cancer Society recognizes virtual colonoscopy screening benefit, March 5, 2008
Copyright © 2009 AuntMinnie.com