The U.S. Food and Drug Administration (FDA) has cleared ClearPoint Neuro's ClearPoint 2.2 Software with Maestro Brain Modeling.
The company is highlighting the first-in-human cases and the publication of a validation study for the software, published in NeuroImage.
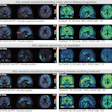
The ClearPoint system uses MRI guidance for peri-procedural segmentation of the cortical structures of the brain to identify both targets and safety zones for cell and gene therapy delivery, laser ablation, biopsy, and deep brain stimulation. The first clinical cases using ClearPoint 2.2 were completed successfully in the first quarter of 2024, and the company expects a full market release in the second half of this year.
The study details the methodology for shape-constrained deformable brain segmentation behind Maestro, describes the validation performed for its FDA clearance, and presents a comparison with manual expert segmentation and FreeSurfer, an open-source segmentation software. Quantitative analysis indicates superior performance compared to both manual expert segmentation and FreeSurfer.