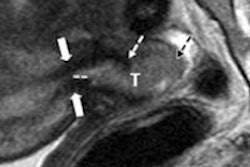
Assessment of tumor response after locoregional therapy with TACE isn't standardized; no universally accepted standard exists, and the number of tumors to assess remains undetermined, said presenter Dr. Vania Tacher of Johns Hopkins Hospital. Current tumor assessment methods are performed on imaging using one-dimensional Response Evaluation Criteria in Solid Tumor (RECIST 1.1) and modified RECIST (mRECIST) and 2D European Association for the Study of the Liver (EASL) measurements after locoregional therapy, but these methods serve as surrogates of tumor volume.
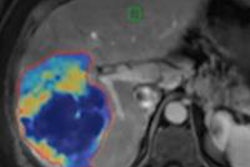
As a result, the researchers sought to examine new 3D tumor volume assessment methods, including the 3D quantitative EASL (qEASL) and volumetric RECIST (vRECIST). They evaluated which methods were best able to assess tumor response in terms of reproducibility, variability, and predictability of patient survival.
The study findings offer a step toward standardization of tumor assessment of HCC following TACE, according to Tacher.
"mRECIST and qEASL are valuable tumor assessment methods -- highly reproducible with a low variability of measurements for predicting survival based on primary index lesion response, target tumor response, and overall response," she told AuntMinnie.com.
The group also found that 3D tumor measurement with vRECIST and qEASL enables estimation of real tumor burden and can help interventional radiologists plan interventions, Tacher said.
"The intraprocedural use of the various [conebeam CT (CBCT)] techniques as dual-phase CBCT helps to see in 3D the tumor burden and to assess treatment success similarly to the pre- and post-TACE diagnostic imaging modalities as MRI or MDCT," Tacher said. "The intraprocedural 3D tumor assessment leads toward a more precise tumor chemotherapy delivery and an early treatment success assessment, which are [predictive of] tumor response on follow-up imaging and survival."