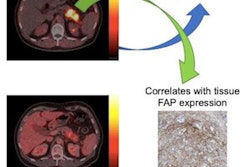
Hybrid PET/MR imaging with an experimental gallium-68 (Ga-68)-labeled fibroblast activation protein inhibitor (FAPI) radiotracer outperforms standard molecular imaging for diagnosing gastric cancer, according to a study published in the January issue of the Journal of Nuclear Medicine.
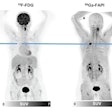
Chinese researchers compared Ga-68 DOTA-FAPI-04 (abbreviated as Ga-68 FAPI) PET/MR and F-18 FDG-PET/CT for diagnosing primary tumors and metastatic lesions in patients with known gastric cancer. They found the experimental radiotracer was better at detecting both primary and metastatic tumors.
"Ga-68 FAPI-PET/MR outperformed F-18 FDG-PET/CT in visualizing primary and metastatic lesions of gastric cancer and may potentially replace F-18 FDG-PET/CT," wrote first author Dr. Chunxia Qin of Huazhong University of Science and Technology in Wuhan.
Gastric cancer is the fifth most common malignant tumor and the third most common cause of cancer death in the world, with many patients presenting with advanced-stage disease because of the lack of early signs and symptoms.
F-18 FDG-PET/CT is widely used in the diagnosis, staging, and preoperative evaluation of gastric cancer. However, F-18 FDG-PET/CT has been reported to have a low detection rate for primary gastric cancer, especially in the early stages, according to the authors.
"Early diagnosis and accurate staging are crucial for choosing an appropriate therapy strategy," the researchers wrote.
Fibroblast activation protein (FAP) is overexpressed in cancerous fibroblasts. Ligands have been developed to inhibit this overexpression (FAPIs) and when labeled with isotopes such as Ga-68 can help visualize tumors on imaging. These FAPI radiotracers appear promising for diagnosing various cancers, including lung cancer, breast cancer, prostate cancer, sarcoma, and head and neck cancer. The performance of FAPI imaging in gastric cancer has been described in only a few case reports, the authors wrote.
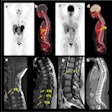
In this prospective study, the researchers recruited 20 patients, nine men and 11 women between the ages of 29 and 70 with biopsy-proven gastric tumors. Each participant underwent both F-18 FDG-PET/CT (Discovery VCT, GE Healthcare) and Ga-68 FAPI-PET/MR (Signa PET/MR, GE Healthcare) scans between June 2020 and July 2020.
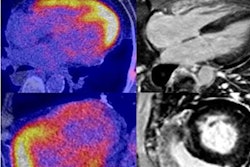
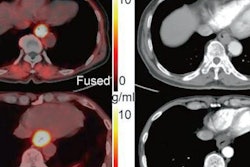
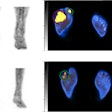
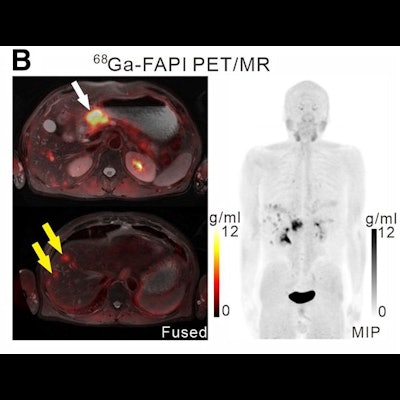
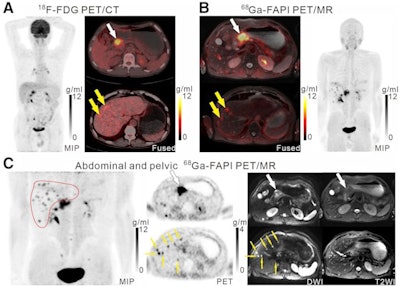 A 61-year-old man with moderately differentiated gastric adenocarcinoma. In addition to the primary tumor (A, white arrow, SUVmax = 11), two foci of elevated activity in the liver were noted on the F-18 FDG PET/CT images (A, yellow arrows, SUVmax = 5.8). On the Ga-68 FAPI PET/MR images, the primary tumor had more intense uptake (B and C, white arrows, SUVmax = 14.2), and the two hepatic lesions had more prominent Ga-68 FAPI accumulation (B, yellow arrows, SUVmax = 7.6). Also, multiple foci of increased Ga-68 FAPI activity were also revealed in the liver (C, red outline, yellow arrows), which corresponded to multiple high signals on diffusion-weighted imaging (yellow arrows), suggesting multiple hepatic metastases. Image courtesy of the Journal of Nuclear Medicine.
A 61-year-old man with moderately differentiated gastric adenocarcinoma. In addition to the primary tumor (A, white arrow, SUVmax = 11), two foci of elevated activity in the liver were noted on the F-18 FDG PET/CT images (A, yellow arrows, SUVmax = 5.8). On the Ga-68 FAPI PET/MR images, the primary tumor had more intense uptake (B and C, white arrows, SUVmax = 14.2), and the two hepatic lesions had more prominent Ga-68 FAPI accumulation (B, yellow arrows, SUVmax = 7.6). Also, multiple foci of increased Ga-68 FAPI activity were also revealed in the liver (C, red outline, yellow arrows), which corresponded to multiple high signals on diffusion-weighted imaging (yellow arrows), suggesting multiple hepatic metastases. Image courtesy of the Journal of Nuclear Medicine.Scans were performed in 14 patients for initial staging and in six patients for the detection of cancer recurrence. The researchers used a visual scoring system and maximum standardized uptake values of the radiotracers in tumors (SUVmax) to compare how the techniques performed, while two groups of experienced nuclear medicine physicians analyzed the results.
The group found that Ga-68 FAPI-PET/MR was superior to F-18 FDG-PET/CT for diagnosing primary tumors (100% vs. 71.43%) and peritoneal metastases (100% vs. 40%) and that Ga-68 FAPI had significantly higher tracer uptake levels (p < 0.05).
In addition, multiple sequences of MR images were beneficial for the interpretation of hepatic metastases in three patients, uterine and rectal metastases in one patient, ovarian lesions in seven patients, and osseous metastases in two patients, according to the findings.
"We found that Ga-68 FAPI-PET imaging was superior to F-18 FDG-PET imaging in detecting primary lesions and metastases in patients with gastric cancer at the initial diagnosis and recurrence detection, as Ga-68 FAPI-PET detected more or larger lesions and showed higher tracer uptake," the researchers wrote.
Ultimately, whether Ga-68 FAPI-PET/MR can be clinically beneficial for the staging of patients with gastric cancer in early stages needs further assessment, the researchers wrote. In this study, tumor staging assessments weren't conducted because most enrolled patients were at an advanced stage, they wrote.
"We should conduct a clinical trial involving a larger number of gastric cancer patients with comprehensive pathologic types and earlier stage to remedy these limitations and further confirm our results," the group concluded.