A tailored breast cancer screening program for women between the ages of 40 and 49 is not only feasible and affordable, it's effective for finding asymptomatic, low-stage tumors, according to a study published in the August issue of Radiology.
The optimal age at which women should begin regular screening mammography continues to be debated, and the question is further confounded by mammography's limitations in imaging dense breast tissue, which tends to be more prevalent in younger women. Researchers are seeking ways to make the most of mammography and catch cancers early by creating screening practices personalized for high-risk women, including additional imaging when necessary, while at the same time keeping radiation dose low.
In this study, Italian researchers used mammography as well as supplemental ultrasound and MRI to evaluate whether a screening program customized for younger women based on their risk factors would improve the cancer detection rate. To reduce lifetime radiation exposure to these younger women, the researchers used a photon-counting mammography system design (MicroDose Mammography, Philips Healthcare) that delivers up to 40% lower radiation dose than conventional full-field digital mammography systems, according to lead author Dr. Elena Venturini, of the San Raffaele Scientific Institute in Milan, and colleagues.
The study included a total of 1,666 women. Before undergoing mammography, each woman filled out a questionnaire that listed possible risk factors for breast cancer (race, body mass index, age at menarche and first child birth, contraceptive and hormonal replacement therapy, menopause, maternal and paternal family history of breast and ovarian cancer, and previous invasive breast examinations, breast cancer, or mediastinal irradiation) (Radiology, August 2013, Vol. 268:2, pp. 347-355).
The researchers assessed the women's breast cancer risk using the Gail test, and after mammography, they developed a screening protocol for each depending on lifetime risk and breast tissue density.
The average lifetime risk of breast cancer was 11.6%. The women's risk was categorized in three ways:
- Women who had low-density breast tissue and a lifetime risk of less than 25% received only mammography.
- Women with negative previous mammograms but dense breast tissue and a lifetime risk of less than 25% and also women with breast implants were classified as having intermediate risk and received ultrasound in addition to mammography.
- Women with a lifetime risk of more than 25%, a genetic risk of breast cancer, or a diagnosis of atypical ductal hyperplasia or lobular carcinoma in situ were classified as high risk and received ultrasound, breast MRI, and mammography.
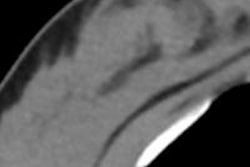
Nine women (0.5%) had a high risk of breast cancer and received all three modalities, while 835 study participants (50.1%) underwent screening ultrasound and mammography, mostly because of high breast density. The rest received only mammography.
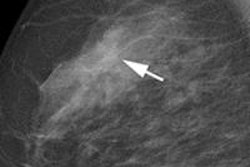
Breast cancer was diagnosed in 14 women across the entire study population, for a cancer detection rate of 8.4 cases per 1,000 women. Twelve of these cancers were found with digital mammography, while two were identified by supplemental ultrasound in women with dense breasts. The 14 cancers proved to be four ductal carcinomas in situ and 10 invasive carcinomas (five of which were stage I).
"The majority of these patients had an excellent or good prognosis," Venturini and colleagues wrote. "Thus, as some recent studies have stated, breast cancer screening in women aged 40 to 49 years enables cancer detection at a lower stage of disease, more frequent conservative treatment, and a reduced rate of recurrence."
MicroDose Mammography performed well in this age group, even in the women with dense breast tissue, with a low average glandular radiation dose of 1.49 mGy, the authors wrote.
The researchers calculated that the total cost to screen all of the women was 115,418 euros ($153,000 U.S.), with a cost of 8,244 euros ($10,928) per breast cancer diagnosed (close to the $10,566 cost reported by the U.S. National Breast and Cervical Cancer Early Detection Program, Venturini's team noted). The cost for a cancer detected with mammography was 6,377 euros ($8,455), while the cost for a cancer detected with only ultrasound was 19,158 euros ($25,401).
Limitations of the study included its small population size, the fact that the group did not investigate the interval cancer incidence or the long-term benefits of breast cancer screening, and the potential for underestimated breast cancer risk using the Gail model, the authors wrote. However, the study findings do indicate that customized breast cancer screening in younger women is effective.
"A tailored breast cancer screening program that uses MicroDose Mammography in 40- to 49-year-old women is feasible and effective and revealed a high cancer detection rate of asymptomatic low-stage tumors with affordable costs, considering the life expectancy of this population," the researchers concluded. "These results support the screening of women aged 40 to 49 years."