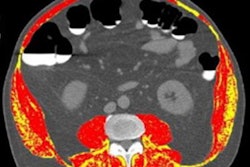
Body composition analysis can be performed on chest CT exams with help from an automated pipeline that includes deep-learning algorithms, according to research presented recently at RSNA 2021 in Chicago.
A multi-institutional research team led by presenter Christopher Bridge, PhD, and senior author Dr. Florian Fintelmann from Massachusetts General Hospital developed a method that yields automated muscle and adipose tissue measurements that correlate highly with manual segmentations.
These results show the potential for body composition analysis -- quantification of the amount and distribution of muscle and fat in the body -- to be performed opportunistically on imaging of areas other than the abdomen, Bridge said.
Much of the previous work in the literature has focused on analyzing a single image slice at the level of the third lower vertebrae (L3). This approach has been used in a variety of applications, including detecting frailty, as well as risk stratification for overall patient survival, surgical complications, cerebrovascular accidents, or myocardial infarct, according to Bridge.
"And several automated pipelines have demonstrated good results in the abdomen, suggesting that it's possible to use body composition-derived metrics as an opportunistic biomarker from routine scans," he said.
However, the focus on the L3 vertebrae limits the applicability of body composition analysis to just studies that contain the L3 slice. To date, there haven't been many efforts to investigate body composition analysis on chest CT scans, despite the ubiquity of those studies, he said.
In a previous effort working with a number of collaborators, the researchers showed that body composition metrics derived from three levels of chest CT images -- T5, T8, and T10 -- are predictive of outcomes such as hospital length of stay and postoperative complications in patients undergoing lobectomy for lung cancer.
Unfortunately, this technique requires tedious, manual segmentation of hundreds of images. As a result, the researchers sought to develop a fully automated pipeline to perform the body composition analysis.
They first assembled a dataset of 629 patients who had axial CT scans prior to lobectomy for lung cancer. The patients were scanned using 36 distinct scanner models from four different manufacturers. The exams included a mix of contrast-enhanced and noncontrast images and had a range of slice thicknesses between 1.25 mm and 5 mm.
After slices with incomplete visualization of the relevant tissues were excluded, the remaining slices were manually segmented by radiologists to extract muscle and adipose tissue compartments.
Of this dataset, 75% were used for training deep-learning algorithms and 10% were used for validation. The remaining 15% were used for testing.
Their pipeline consists of two independent neural networks. After receiving a series of images, the first model selects a single image that's representative of each of the T5, T8, and T10 vertebral levels. Next, a segmentation network separates the image into skeletal muscle and adipose tissue compartments.
In testing, the trained system was highly accurate for quantifying tissue cross-sectional areas.
| Performance of automated pipeline for body composition analysis on chest CT scans | ||||
| Cross-sectional area | Median attenuation | |||
| Muscle | Adipose tissue | Muscle | Adipose tissue | |
| Median absolute error | 3.1 cm2 | 4.6 cm2 | 1.0 Hounsfield units (HU) | 1.0 HU |
| Intraclass correlation coefficient | 0.985 | 0.994 | 0.969 | 0.992 |
"We found that most of the disagreement was actually not disagreement in the segmentation but small variations in the exact slice that was selected for analysis, which can have a relatively large effect on the area of the tissue," he said.
Bland-Altman analysis also showed consistently accurate quantification and attenuation across all three vertebral levels and the two different tissue types, he said.
"And this [automated method] enables large-scale biomarker collection or analysis of patients," he said. "It also enables us to automate prediction of hospital length of stay for lung lobectomy that we previously demonstrated, and possibly many other things."