Medical image quantification software developer Quantib has received U.S. Food and Drug Administration (FDA) clearance for its Quantib Neurodegenerative (ND) artificial intelligence (AI)-based software.
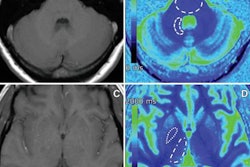
Designed to support radiologists in interpreting brain MRI scans, the software measures brain atrophy and detects white-matter hyperintensities, according to the vendor. These changes in the brain are related to, for example, aging, dementia, and multiple sclerosis, Quantib said.
Quantib ND performs fully automatic segmentation of lobes and the hippocampus to objectively assess the development of atrophy. In addition, it provides segmentation of white-matter hyperintensities for monitoring neurological changes in these patients, according to the firm. It also offers reference centile curves derived from the Rotterdam Scan Study.
Neurodegeneration over time can also be tracked via Quantib ND's follow-up feature, while an editing capability enables radiologists to have full control over the software's results before sending them to the PACS, Quantib said.