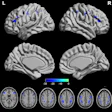
With all the scientific debate in recent years over the long-term effects of gadolinium retention after contrast-enhanced MRI scans, two patients are leading a campaign to advance research on the topic and, most importantly, ensure that those adversely affected by gadolinium are heard and not forgotten.
Sharon Williams and Hubbs Grimm created the Lighthouse Project in 2014 to increase awareness among patients and physicians of how gadolinium retention can have lingering, devastating effects. The ailments might not quite compare with the most severe consequences such as nephrogenic systemic fibrosis (NSF), but the pain and debilitation these patients experience is very real.
"We see the Lighthouse Project as providing factual information and a voice for those patients who so far have not been heard," Williams said. "Many go to doctors and try to get them to listen and to help them. But [the doctors] don't know what to do because, let's face it, there is not a cure for this. I think sometimes doctors don't realize how important it is to a patient just to have them listen to them and believe them."
Gadolinium experiences
Williams met Grimm in 2011, while they both were experiencing health problems they believe were related to receiving contrast for MRI scans. Grimm has had nine contrast-enhanced MRI scans, and the last three were performed within a two-week period. One month later, his toes started to become numb and then began tingling.
 Hubbs Grimm, co-founder of the Lighthouse Project.
Hubbs Grimm, co-founder of the Lighthouse Project."Some of the symptoms caused the doctor to order more contrast MRIs for symptoms that have been caused by the gadolinium in the earlier MRIs. I did not know any better," he said. "The reality was the more contrast MRIs he gave me, the worse it got."
Grimm since has been diagnosed with small fiber neuropathy, which can be caused by metal toxicity and/or diabetes. After a series of tests, including an electromyogram and CT scan at the Cleveland Clinic, Grimm said clinicians termed his situation "idiopathic, which means they don't know why it happened."
Because metal toxicity is a possibility, Grimm has tracked his urine test results since 2010; the gadolinium levels in his discharge are above normal, especially after 7.5 years. Even chelation treatments haven't helped.
Williams had a series of five MRI scans beginning in 2010. Except for symptoms that might be related to her Hashimoto's thyroiditis, she said her doctors have not found any cause for her pain, worsening vision, and thickening tissues. Testing for all autoimmune diseases and scleroderma were negative.
Subsequently, part of her thyroid gland was removed in 2014. At that time, Dr. Thomas Darrah at Ohio State University tested the tissue and found a significantly elevated concentration of gadolinium. He attributed the elevated levels to a "unique gadolinium-rich source consistent with gadolinium-based contrast agents," Williams said.
(Darrah led a November 2009 study that documented gadolinium retention in the bone tissue of patients exposed to chelated gadolinium-enhanced MRI scans [Metallomics, November 2009, Issue 6, pp. 479-488].)
When Williams met Grimm six years ago, they were both members of a now-defunct gadolinium-related support group. In 2012, they began working together on gadolinium-related projects through a new Yahoo-based group. That's also when Williams secured the domain name of GadoliniumToxicity.com, with the thought that the duo might want to create their own website someday.
Support group
The Lighthouse Project is the conduit for a support group that currently has approximately 230 members from all walks of life and age groups. The majority of members are from the U.S., while other participants are from countries such as Canada, Australia, Spain, the U.K., China, Malaysia, and Iran.
 Sharon Williams, co-founder of the Lighthouse Project.
Sharon Williams, co-founder of the Lighthouse Project.The co-founders ask prospective members to undergo a 24-hour urine test for gadolinium. Members can then send copies of the results to Grimm, along with the date of their last contrast MRI and the number of MRI scans they have had. Grimm, who is quick to tell the members he is not a medical professional, compiles the data and tells the member how he or she compares with peers in terms of the test results.
Williams and Grimm also screen people when they apply for membership to avoid those who might be trolling for patient data or who may have similarly unscrupulous motives. As an example, Williams cited problems experienced by members of NSF groups, who were targeted by the legal community.
"What is frustrating for me is the fact that we are getting so many young adults. I hate to think that these young people in their 20s and early 30s could be dealing with this for the rest of their lives," she said. "I've had two individuals who contacted me who are suicidal. It is about the fear of the unknown. What we know about NSF is the greatest fear, especially of the younger patients, who in some cases react much more severely to the gadolinium in their bodies than others do."
Professional collaboration
Partly due to its collection of data from members, the Lighthouse Project has drawn attention from several researchers focusing on gadolinium-based contrast agents (GBCAs).
The co-founders credited Dr. Emanuel Kanal, director of MR services at the University of Pittsburgh, with providing support and input for the Lighthouse Project's first survey in 2014. The self-published paper reported on symptoms of patients with elevated levels of gadolinium in their urine and common chronic symptoms of gadolinium toxicity that were consistent with NSF.
The group has also provided information for research by Dr. Richard Semelka, director of MR services and a professor of radiology at the University of North Carolina at Chapel Hill. Some Lighthouse Project members have been subjects in Semelka's surveys on GBCAs. In May 2016, Semelka was the lead author of a paper proposing new terms to describe aftereffects of gadolinium exposure that fall short of NSF.
Last month, Williams and Grimm published their most recent paper on the website, in which 70 cases were analyzed involving 120 tests for gadolinium in 24-hour urine samples. The collections were taken as few as four days to as many as 10 years after contrast administration. The subjects all believe they have symptoms of gadolinium toxicity caused by contrast-enhanced MRI scans.
In 21 cases, urine testing was performed in the first month following a GBCA-enhanced MRI scan. The samples showed gadolinium in the urine as long as four months later, meaning the contrast had not cleared the body in a few days, as most medical practitioners believe.
In addition, there were eight cases in which an individual underwent urine testing more than three years after his or her last contrast-enhanced MRI. Levels of gadolinium appeared to be related to the number of scans that each patient received, which ranged from one to nine.
While the authors noted that their methods "do not meet the rigor" of a conventional clinical trial, they contend the findings "present a strong case that gadolinium from contrast-enhanced MRIs may be present in patients with normal renal function for considerably longer than would be expected based on the elimination rates reported in GBCA product labeling."
Giving due credit
Williams and Grimm likened their results to those of a 2016 study by Semelka et al, which found evidence of gadolinium among 42 subjects with normal renal function and no pre-existing clinical symptoms more than 30 days after a contrast-enhanced MRI scan. A multitude of patients also reported central, bone, and peripheral pain and headache (Magnetic Resonance Imaging, December 2016, Vol. 34:10, pp. 1383-1390).
Both studies' findings "indicate stronger action is needed by the [U.S. Food and Drug Administration (FDA)] and others to inform patients about possible gadolinium retention from contrast-enhanced MRIs and the potential for side effects," Williams and Grimm wrote.
"Give credit where credit is due," said Tobias Gilk, senior vice president of Radiology-Planning, founder of Gilk Radiology Consultants, and an MRI safety advocate. "This self-organized patient community has done more to shine a light on the questions and concerns that I feel have not been answered by the scientific and professional communities about their contentions of injuries associated with gadolinium."
Even followers "with a bucket full of skepticism" on the gadolinium issue should look at the assembled data from the group on the "antagonized release of gadolinium from their bodies weeks, months, and years following the administration," Gilk added.
Future plans
Williams plans to continue her work with the Lighthouse Project, but she hopes the number of people joining the support group will slow down, as the initiative occupies a great deal of her time.
"My goal is to do whatever I can, to talk to whomever, and to work with people in radiology or elsewhere in medicine to try to figure this out, so that something is done," she said. "I am hoping that at some point in time I can close the book on this and go back to living the life, but I don't see that happening for a while."
Grimm also plans to continue his part in the effort to gather evidence on the effects of gadolinium retention and to provide a place where people can learn more if they think they are gadolinium-toxic.
"People are coming here, so it is important for us to be there and try to provide as much information as we can, so they can look at it, get a feeling for where they are, and know where they might go with what's going on," Grimm said. "I am an eternal optimist. My assumption is that someday we will make progress."