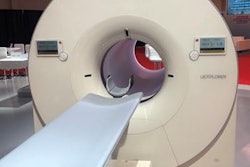
Shanghai-based United Imaging Healthcare (UIH) said it has received U.S. Food and Drug Administration (FDA) clearance for uExplorer, a total-body PET/CT scanner developed in collaboration with researchers from the University of California, Davis (UCD).
The system can capture a 3D image of the whole human body in one position, according to the vendor. Simon Cherry, PhD, a professor of biomedical engineering at UCD, and Ramsey Badawi, PhD, a professor of radiology, unveiled the first images from the device in November, and a mock-up of the system was shown in United's booth at RSNA 2018.
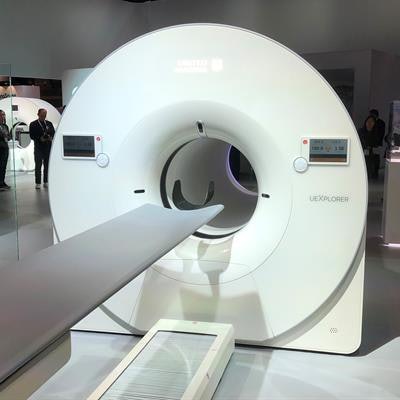
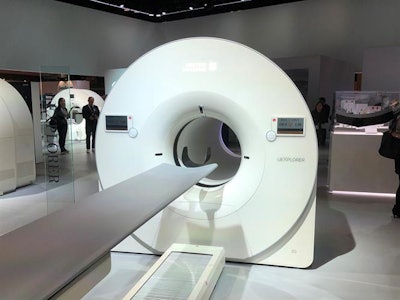 UIH displayed a mock-up of uExplorer at RSNA 2018.
UIH displayed a mock-up of uExplorer at RSNA 2018.Early versions of the scanner developed by UCD featured a half-million PET detectors lining the bore of the system. The configuration is believed to result in less radiation delivered to patients because the system acquires almost all the available signal from the radiotracer.
In addition, researchers believe that uExplorer could reduce total-body scan times down to one minute. This could be particularly beneficial for imaging pediatric patients without anesthesia or sedation, as well as adult patients who cannot withstand prolonged scanning.
UIH said it expects the technology will be useful for improving diagnostics, tracking disease progression, and researching new treatments. The unit could be used, for example, to better visualize cancer that has spread beyond a single tumor site, measure blood flow across the entire body, and study inflammation, infection, and immunologic/metabolic disorders, according to the firm.
Commercial availability in the U.S. is expected early this year.