SNM has created the Molecular Imaging Clinical Trials Network (MICTN) to streamline processes for utilizing imaging biomarkers in clinical research and clinical practice.
The Reston, VA-based organization has designed a model for the use of imaging biomarkers in clinical trials that spans drug development, molecular imaging, radiolabeled probe development, and manufacturing and regulatory issues.
The network also is intended to provide centralized investigational new drugs (INDs) for biomarkers of interest to the pharmaceutical and imaging communities, as well as coordinate standardized imaging protocols across qualified multicenter clinical trial sites.
The network also will provide information on qualified radiopharmaceutical manufacturers to help design and develop clinical trials.
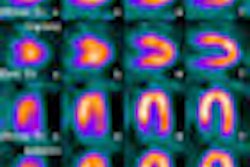
The network received approval for the first centralized IND in August for F-18 fluorothymidine (FLT), an investigational PET imaging biomarker that has shown promise in the demonstration of tumor proliferation as a surrogate marker of effectiveness in the development of novel cancer therapies.
Several pharmaceutical developers have already expressed interest in utilizing this approved IND in near-term clinical trial work. Active clinical trials utilizing FLT are expected to begin in 2009.
Related Reading
SNM has 'serious concerns' over isotope situation, August 28, 2008
SNM draft report shows U.S. Mo-99 production years away, July 22, 2008
SNM cites regulatory, reimbursement, and research as top priorities, June 18, 2008
SNM's Images of the Year show molecular imaging's head-to-toe prowess, June 16, 2008
Nuclear medicine must overcome obstacles to flourish in the future, June 15, 2008
Copyright © 2008 AuntMinnie.com