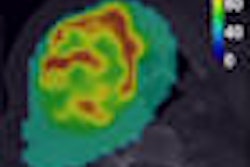
Siemens Healthcare of Malvern, PA, is touting recent clinical results of a new PET biomarker for imaging hypoxic tumors.
The study was presented last week at the World Molecular Imaging Congress in Nice, France, and details results produced with HX4, a new PET biomarker being developed by the company's Siemens Molecular Imaging Biomarker Research facility in Los Angeles.
Siemens researchers working in collaboration with a team from Fox Chase Cancer Center in Philadelphia found HX4 to be stable for imaging at 145 minutes after injection, that it would safely clear the body through urinary elimination, and that there were very low-dose accumulations in major organs.
Hypoxic cells are clinically problematic and tend to be less responsive to standard treatment regimens, according to Siemens. The development of an imaging biomarker that selectively identifies hypoxic tumor cells could help radiation oncologists tailor specific treatment options to most efficiently manage disease, the company believes.
Siemens intends to distribute HX4 through its PETNet Solutions radiopharmacy business.
Related Reading
Siemens adds to preclinical imaging line, September 10, 2008
FDA clears Siemens Ysio wireless DR, September 9, 2008
Siemens plans MA expansion, September 2, 2008
Siemens sends layoff notices, August 18, 2008
Siemens completes Northampton installations, August 12, 2008
Copyright © 2008 AuntMinnie.com