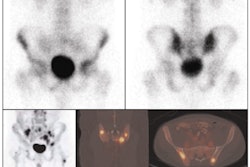
GE Healthcare has received U.S. Food and Drug Administration (FDA) clearance for a new PET/CT scanner.
The system, which has yet to be named, will be optimized for use in oncology applications, according to the Chalfont St. Giles, U.K.-based vendor. It features GE's high-sensitivity crystals along with its Vue Point HD high-definition image processing application and motion-management techniques for improving the detection of lesions, treatment planning, and quantitative accuracy, GE said.
The first shipments of the system are expected to begin in the fourth quarter of 2008.
Related Reading
GE moves Optison agent through EU regulation, July 23, 2008
Dineen replaces Hogan at GE Healthcare, July 17, 2008
GE adds charges for MRI scanners, July 15, 2008
OEC shipments drive GE's Q2 growth, July 11, 2008
GE donates CT system for 2010 Olympics, July 10, 2008
Copyright © 2008 AuntMinnie.com