Breast Tumors:
Age is a strong predictor of breast cancer survival, and young age at the time of diagnosis is an independent risk factor for a poor prognosis [112].
Mammography and breast ultrasound are the modalities primarily used to screen for breast cancer. Mammography is highly sensitive and can identify 80-90% of patients with breast cancer [2]. Unfortunately, a positive mammographic finding does not always equate with malignancy. Only between 2 to 4 of 10 patients with abnormal mammograms are found to have breast cancer at histologic analysis [2]. Additionally, about 10% of breast cancers cannot be identified mammographically even when they are palpable [2]. Invasive lobular carcinoma can be difficult to identify mammographically because of its diffuse growth pattern and tendency to have an opacity equal to or less than the surrounding parenchyma [2]. Ultrasound can permit rapid differentiation of cystic and solid lesions, but accurate differentiation between benign and malignant solid lesions is sometimes difficult [2].
Invasive ductal carcinoma is the most common type of breast cancer (70-80% of cases), while invasive lobular carcinoma is relatively uncommon (5-15% of cases) [44,84]. Almost all breast cancers are FDG-avid [82], but display a considerable variation in FDG uptake [7]. Invasive ductal carcinoma exhibits significantly higher FDG uptake compared to invasive lobular carcinoma [7,84]. Focal nodular lesions also show higher accumulation when compared to infiltrative/diffuse lesions [7]. Additionally, the greater the degree of tumor proliferation and the greater the degree of tumor dedifferentiation, the greater the FDG accumulation [7]. The estrogen and progesterone status of a lesion do not appear to influence FDG uptake [7], but other authors report higher FDG uptake in ER negative and triple negative breast cancers [82]. Multiple other factors influence uptake of FDG in breast cancer cells including the microvasculature for delivering nutrients, Glut-1 surface receptors for transport of FDG into the cell, hexokinase levels for entering FDG into glycolysis, the proliferation rate of the tumor, and the level of hypoxia-inducible factor-1-alfa for upregulating Glut-1 receptors [17].
HER-2 positive tumors (human epidermal growth factor receptor type 2 tumors) account for 15-20% of total cases [114]. These lesions respond to HER2-targeting therapeutics, such as the HER-2 targeted monoclonal antibodies trastuzumab and pertuzumab, the tyrosine kinase inhibitor lapatinib, or trastuzumab-based antibody-drug conjugates [114]. Note that metformin therapy has been shown to decrease human epidermal growth factor receptor (HER2) membrane density (metformin induces HER internalization) and depletion of cell-surface HER decreases antibody tumor binding for imaging and therapeutic purposes [120]. Metformin alters the cholesterol content located at the cell membrane and intracellularly, and it affects the synthesis and stability of receptors that rely on GM1 ganglioside, lipid raft markers (such as HER) [120]. The effect of metformin is reversible and after the agent is discontinued, antibody tumor binding is restored [120]. Interestingly, patients with metabolic dysregulation who take metformin have a lower cancer risk and cancer related mortalities [120]. Metformins antitumoral effecrs are likely related to it's inhibiting the phosphatidylinositol-3-kinase mammalian-target-of-rapamycin and Ras-mitogen-activated protein kinase (MAPK) pathways [120].
Triple-negative breast cancer (TNBC) accounts for 15-20% of invasive breast cancers and is characterized by the lack of estrogen and progresterone receptors and the absence of HER2 overexpression (it is therefore unresponsive to the usual endocrine therapies) [58,61,85]. Triple-negative breast cancer more frequently affects younger women with a family history of breast cancer [85]. triple-negative breast cancer accounts for up to 25% of breast cancer diagnoses in women younger than 40 years [112]. Premenopausal African American women also have a higher risk of developing triple receptor negative breast cancer [61] as do patients with BRCA1 mutations [98]. It has a more aggressive clinical course than other forms of breast cancer (in part related to high nuclear grade (> 80% grade 3) and absence of effective targeted therapy) and patients with triple negative breast cancer have a relatively poor outcome with a higher relapse rate (which peaks 1-3 years after treatment) [58,61,85,112]. Patients with triple-negative breast cancer also have a high early incidence of brain metastases [61]. However, patients that have a complete pathologic response to neoadjuvant therapy have a good prognosis with a 3 year overall survival of 94% (compared to 68% for patients with residual tumor at the time of surgery) [58]. Chemotherapy is the mainstay of neoadjuvant therapy for patients with TNBC, but the addition of pembrolizumab (for patients with PDL1- positive tumors, assessed by a combined positive score of >/= 10%) has been associated with improved PFS [122].
Invasive lobular carcinoma (ILC) accounts for about 15% of breast cancers, but is more frequently multifocal and bilateral compared to invasive ductal carcinoma [119]. The lesion is more frequently occult on mammography, ultrasound, and MRI [119]. FDG PET also has a lower sensitivity for ILC because of lower tumor microvascularity, cellular density, proliferation rate, and levels of glucose transporter proteins [119]. ILC lesions have a high rate of ER positivity- approximately 95% of lesions exhibit ER expression [119]. FES PET may be a better agent for imaging ILC- in one study, FES OET demonstrated abnormal uptake in 83% of sites of histologically known ILC, depicted additional lesions in 24% of patients compared to standard of care, and the exam findings changed the clinical stage in 18% of patients [119]. Limitations of FES imaging include very high liver activity which will obscure lesions and FES PET cannot be performed in patients undergoing systemic therapy with ER modulators or degraders [119].
Inflammatory breast cancer is a rare (1-2% of primary breast cancers), but aggressive, rapidly proliferating form of breast cancer [45,79]. The criteria for the clinical diagnosis includes erythema, edema involving more than 2/3's of the breast, peau d'orange changes, breast enlargement, warmth, tenderness, and breast induration on palpation [45]. About 20% of patients with IBC have distant metastases at the time of diagnosis [45]. The standard treatment is neoadjuvant chemotherapy followed by modified radical mastectomy and subsequent radiation therapy [45]. The mean 5 year survival following multidisciplinary therapy is 20-40%, with a median survival of 12-36 months [45,79].
Tumor genetics/associated syndromes:
BRCA mutations:
BRCA1 and BRCA2 are large tumor suppressor genes located on chromosomes 17 and 13, respectively [98]. These genes encode for proteins that maintain genomic stability through DNA repair and transcriptional regulation [98]. Cells that have harmful BRCA mutations are unable to repair DNA damage that results in accumulation of chromosomal abnormalities leading to uncontrolled proliferation and tumor development [98]. The BRCA gene mutation can be inherited from either parent in an autosomal dominant pattern [98]. Woman with a BRCA mutation have a substantially greater risk if developing breast cancer [98]. About 5-10% of breast cancer patients have a BRCA mutation [97,98,105], however, 80% of patients with the mutation will develop breast cancer (and up to 50% will develop ovarian cancer) [97]. Other authors suggest that 55-65% of females with BRCA1 mutation and 45% of those with BRCA2 mutation will develop breast cancer by age 70 years [98].
Women with the BRCA1 mutation are repoprted to develop breast cancer at an early age (average age at diagnosis for breast cancer in BRCA1 patients is 40 years compared to 61 years for the general population [98]) [61]. Additonally, BRCA mutation carriers have an up to 63% risk of developing a contralateral primary breast cancer [98]. Other authors state that BRCA mutation carriers have a 7-17% incidence of a second breast cancer after initial diagnosis and this risk is highest in BRCA1 carriers who received their initial diagnosis when they were younger than 40 years of aege [112]. Breast cancers in patients with BRCA1 mutation are more often triple receptor negative (up to 69% of cases), high grade, and poorly differentiated [98,105]. BRCA2 associated breast cancers are similar to sporadic lesions with more than 75% being estrogen receptor positive and only 16% being triple negative [98].
The majority of cancers associated with BRCA mutations are invasive ductal carcinomas [105]. Medullary breast cancer accounts for less than 1% of sporadic malignancies, but are seen in up to 19% of BRCA1 carriers, and approximately 15% of patients with medullary breast cancer carry the BRCA1 mutation [98]. Ductal carcioma in situ is common in BRCA2 mutation carriers [105]. Additonally, BRCA2 associated cancers tend to be estrogen receptor positive [105].
Ovarian cancer is more common in BRCA1 mutation carriers (lifetime risk 40%) than in BRCA2 carriers (11-17% lifetime risk) [105]. BRCA1 mutation carriers younger than 50 years have a fivefold relative risk for developing colon cancer [105].
Cowden syndrome:
Cowden syndrome is an autosomal dominant hereditary cancer syndrome that results from a mutation in the PTEN (phosphate and tensin homolog) gene [103]. The PTEN gene encodes for a regulator of the phosphoinositide 3-kinase/protein kinase B and mammalian target of rapamycin pathways that function in cell cycle regulation, proliferation, and apoptosis [103]. Clinical features include macrocephaly, trichilemmomas (hamartomas of the outer hair root), acral keratoses, facial papules, and oral papillomas [103]. Thyroid disease occurs in up to 75% of patients including MNG, Hashimotos, and thyroid adenoma [103].
Breast cancer is the most common malignancy associated with Cowden syndrome with a lifetime risk of 25-50% [103]. Other cancers include endometrial cancer (overall risk 5-10%), thyroid cancer (3-10%), renal cell cancer (up to 34% and associated with the papillary subtype) [103].
Focal breast lesions:
One potential benefit of PET imaging is that it is not affected by the density of breast tissue. Image quality is also not impaired by prior surgery, prior XRT, or breast implants [2]. Both SUV analysis (variable cutoffs reported) and visual assessment of lesion uptake have been applied to image interpretation. Reported sensitivities for FDG PET range from 64% to 96%, and specificity's from 75% to 100% [1,2,4,19,21,23]. The positive predictive value is 81-100% and the negative predictive value is 52-89% [47]. In a meta-analysis of 606 patients, PET had a sensitivity of 88%, and a specificity of 79% for differentiating benign from malignant lesions [23]. The relatively high sensitivity in likely due to the large size (2 to 4 cm) of the lesions studied [23]. In a direct comparison with Tc-MIBI imaging, FDG-PET performed similarly for the proper classification of focal breast lesions [4]. Although PET imaging using 18-FDG has demonstrated potential in the differentiation of benign from malignant breast lesions, it is not recommended for routine use in the diagnosis of breast cancer [40].
The primary limitation of FDG-PET imaging for evaluation of breast carcinoma is its inability to detect small lesions (under 1 cm in size) due to partial volume effects and natural tracer accumulation in the breast which may obscure low uptake lesions [2]. The sensitivity of PET is only 25% for tumors smaller than 1 cm, but can be as high as 84% for tumors between 1-2 cm [31]. Although the sensitivity is generally higher for lesions greater than 1 to 2 cm in size, even larger lesions may not necessarily demonstrate tracer accumulation (up to 20% of lesions between 2 to 5 cm in size may be falsely negative on PET FDG imaging) [2].
In general, for breast cancer, FDG uptake intensity correlates with tumor grade and tumor proliferation index (Ki67 expression) [66]. PET is of limited use for identifying tumors that are of well-differentiated histology [37,54]. For a given size tumor, infiltrating ductal carcinoma has higher FDG uptake than infiltrating lobular carcinoma [66]. The reason a large percentage of invasive lobular carcinomas are not detected (65% in one study), is possibly due to low tumor cell density, its infiltrative nature, and low metabolic activity [2,14,54,66]. Tubular carcinoma and ductal carcinoma in situ also show less FDG accumulation and these lesions may not be reliably distinguished from normal breast tissue [10]. Estrogen receptor negative and triple-negative tumors show higher SUV [66]. Most authors have found no correlation between FDG uptake and HER2/neu expression [66]. There may be a correlation between the intensity of FDG uptake and p53 satus [66].
None-the-less, PET imaging may be useful in a carefully selected subgroup of patients with inconclusive conventional imaging procedures. For lesions of sufficient size, a positive PET scan carries a high positive predictive value that the lesion is breast cancer [2]. False-positive findings are also infrequent (about 5% of benign lesions will demonstrate tracer uptake) [2]. False positive exams have been reported in association with inflammation (mastitis) fibroadenomas (about 10% accumulate FDG [2]), and ductal adenomas [2].
Prone imaging and dual time point imaging have been reported to increase the sensitivity and accuracy for the identification of malignant lesions [36,43]. Malignant breast lesions will show a gradual increase in FDG accumulation over time with improved contrast between the lesion and surrounding background, while benign lesions show decreased uptake over time [36,43]. In one study, dual time point PET/CT showed an overall sensitivity of 80%, specificity of 100%, and accuracy of 84% versus 62%, 100%, and 69%, respectively, for single-time point PET [43]. Accuracy and sensitivity was higher for lesions greater than 1 cm [43]. One drawback of dual point PET/CT imaging is the increased patient radiation exposure from the second CT exam [43].
Specialized dedicated PET mammographic imaging systems (PET Emission Mammography or PEM) may hold future promise due to higher resolution (1.5 mm in-plane) [8,9,42,43,63], however, at the present time, FDG PET imaging does not have a clinical role in detecting primary breast cancer [38].
|
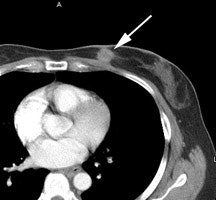
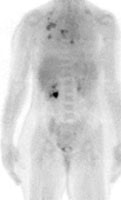
Left breast cancer: The FDG PET exam below is from a patient with a focal 2 cm left breast carcinoma. Note the intense FDG accumulation within the lesion. |
|
|
|
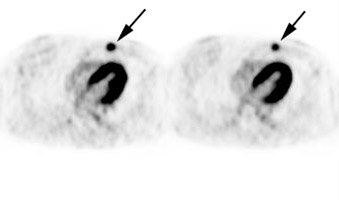
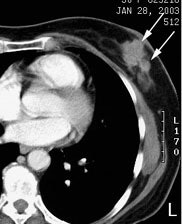
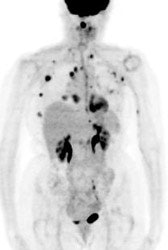
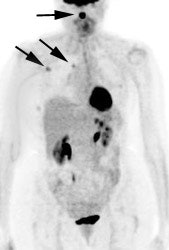
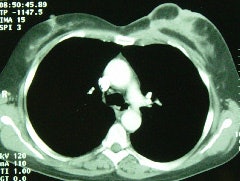
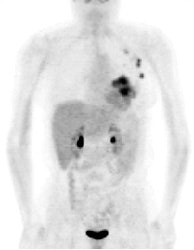
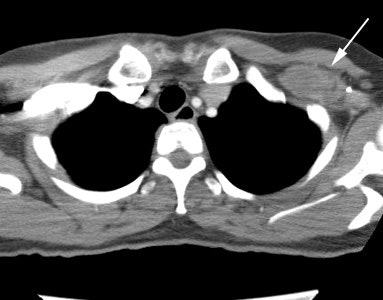
Multifocal breast cancer: The FDG PET exam below is from a patient with a large left breast cancer. The PET exam demonstrated several sites of tracer uptake in the left breast (black arrows). Retrospectively, multiple tumor foci could be identified on the CT scan (white arrows). The presence of multifocal breast cancer can significantly alter patient management. |
|
|
Staging, prognosis, and impact on management:
FDG PET imaging is generally not routinely used in the staging work-up of patients with stage I or early stage II disease due to a low rate of upstaging [45,102]. PET is primarily used for the evaluation of patients with advanced stage breast cancer- including those with a primary tumor larger than 5 cm, skin or chest wall involvement, fixed axillary nodes, positive supraclavicular, infraclavicular, or IM nodes, and patients with inflammatory breast cancer (which is associated with a high rate if distant metastases) [47,66]. Locally advanced breast cancer (LABC) is associated with a high risk of distant metastases and includes N2, N3, or T4 disease - clinical stages IIIA (excluding T3N1), IIIB, and IIIC [65,86]. FDG PET imaging can significantly impact on patient management- particularly in patients with suspected advanced stage disease [11]. Some authors also suggest a role for PET imaging in patient's under the age of 40 years due to the higher risk for ore aggressive tumors [77]. PET imaging may also be beneficial in certain patients with stage IIB cancer- up to 17% of patients under the age of 40 years with stage IIB breast cancer (T2N1/T3N0) can have unsuspected metastatic disease [77]. Other studies report distant metastatic disease on PET/CT in 11-15% of patients with stage IIB and 18-47% with stage III [87,94]. (AJCC Breast Cancer Staging)
PET findings can result in a change in the patients clinical stage in up to 36% of patients (28% upstaged, 8% downstaged) [11]. Unsuspected sites of nodal or distant metastases can be found in up to 20% of patients [11]. In one prospective study of 48 patients with locally advanced breast cancer, PET imaging detected unsuspected distant metastatic disease in 8% of patients (95% CI: 4%-22%). In a prospective study of patients with LABC, PET imaging resulted in a change in stage in 52% of patients and detected metastatic disease not identified on conventional imaging in 35% of patients [65]. In another study of patients with inflammatory breast cancer, FDG PET/CT detected unsuspected sites of distant metastatic disease in 17% of patients [45]. Recent studies have also suggested an important role for PET imaging in the staging of patients with Stage IIB and IIIA disease [54]. In a prospective evaluation, PET imaging modified staging in almost 15% of patients with stage IIB disease, and in almost 28% of patients with stage IIIA disease- particularly women with N2 disease (56% stage modification) [54].
Equally important is the fact that the patient's clinical management can be changed in up to 58% of patients based upon the results of the PET study [11]. For patients with breast cancer, some studies sugegst that tumor SUVmax has been shown to have a strong relationship for prediction of tumor progression and disease free survival, although this is not supported by other studies [65,88]. Other authors suggest that the pretreatment SUVmax of axillary nodal metastases is the most important predictive factor for disease free survival [62]. Finally, total metabolic tumor volume may be a more accurate predictor of overall survival compared to SUVmax [80].
Axillary lymph node evaluation: LN metastasis is the most important predictor of overall recurrence and surivial [104]. Patients with disease localized to the breast have a 5-year survival of almost 99%, while this drops to 86% for patients with regional LN metastases [104]. Pre-operative determination of node status is essential as clinical evaluation is usually inaccurate. Up to 40% of patients with a clinically negative exam demonstrate the presence of metastases at histologic examination. Conversely, 40% of patients clinically suspected of having lymph node metastases, are found to be negative on histologic review [1]. In patients with early stage (T1 or T2) breast cancer and clinically node-negative disease that are subsequently found to have positive SLN, additional axillary node dissection in these patients offered no significant advantage in local or regional recurrence rates compared to patients with SLN biopsy alone (although nearly 0ne-third of the group with axillary node dissection was found to have additional axillary node disease) [113].
Reported PET sensitivities for locoregional nodal metastases range from 50% to 100% [2-5,19,42,66]. Specificity for the detection of axillary nodal metastases ranges from 88% to 96% [19,20,66]. Lower sensitivities for axillary nodal metastases can be expected for metastatic foci smaller than 1 cm in diameter [4] and those with micrometastases [10]. In a prospective study of 360 patients with newly diagnosed breast cancer PET had a sensitivity of 61%, a specificity of 80%, a PPV of 62%, and a NPV of 79% for the detection of axillary nodal metastases [38]. Patients with false negative PET results tend to have smaller and fewer tumor-positive lymph nodes than true-positive cases [47]. Consequently, PET imaging is not recommended for axillary staging and a negative FDG PET exam should not preclude lymphoscintigraphy and surgical axillary node dissection [4,38,66]. Although PET imaging cannot replace sentinel node biopsy, the presence of axillary node uptake is highly suggestive of malignancy (PPV of greater than 80% in most studies) [66]. Ultrasound has also been used to evaluate for axillary node metastases- suspicious features include asymmetric cortical thickening or lobulations, loss of the normal fatty hiuls, abnormal shape (rounded appearance), and increased peripheral flow [47].
|
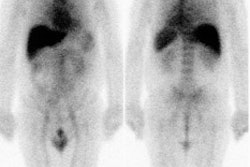
Breast cancer: The patient below was a 44 year old female who had complained of right breast fullness and discomfort from an axillary nodule. An initial mammogram was negative. Over the following six months two additional mammograms were performed and were both negative. A fine needle biopsy of the axillary nodule was subsequently performed and demonstrated carcinoma. An ultrasound guided biopsy of an 8 mm breast lesion was negative. A FDG PET exam was then performed and revealed extensive metastases to axillary and supraclavicular lymph nodes, as well as the primary lesion in the right breast (black arrow). Scan courtesy of the Northern California PET Imaging Center, and CTI. |
|
|
Although PET imaging cannot be used to replace sentinel lymph node dissection, in patients with known axillary metastatic disease, PET imaging can provide additional prognostic information [62]. Higher tracer uptake within axillary lymph node metastases is associated with a greater risk for disease recurrence [62]. A pre-treatment nodal SUV max of 2.8 or less has a sensitivity of about 92% and a specificity of about 87% for predicting disease free survival (in one study disease recurrence was seen on oly 2% of patients with pre-treatment nodal SUVmax below 2.8) [62].
Internal mammary lymph nodes: Studies have shown that FDG PET/CT can detect extra-axillary lymph node involvement in almost one-third of patients with stage II-III disease [69]. Patients with IM nodal involvement have a worse prognosis and decreased overall survival [69,81]. The presence of IM node metastases can upstage disease and alter therapy to include adjuvant chemotherapy or modification of radiation therapy (such therapy has been shown to be associated with improved survival) [69]. Positive IM nodes can be seen in 9% of patients during staging and restaging for breast cancer and is associated with a high likelihood for malignant involvement [69]. Other studies have indicated IM nodal involvement can be seen even without axillary involvement in 6-16% of patients [69]. Risk factors for having positive IM nodes on PET/CT include inner quadrant tumor, increasing tumor size, high tumor grade, the presence of lymphovascular invasion, positive axillary nodes, decreasing patient age (under 50 years), and triple-negative hormonal receptor status [69,81].
Metastatic disease: FDG PET imaging is recommended in addition to conventional imaging for the detection of metastatic breast cancer [40]. The prevalence of distant metastases increases with increasing tumor stage [37]. It is relatively uncommon for patients with Stage I or II disease at the time of diagnosis to have distant metastases [37]. However, for patients with more advanced stage disease (locally advanced breast cancer - LABC- defined as primary breast cancer exceeding 5 cm, fixed axillary lymph nodes, or skin or chest wall invasion [38]), FDG PET imaging can aid in the detection of metastases to lymph nodes and other sites of disease unrecognized by conventional imaging [15,42]. FDG PET imaging has been shown to be superior to conventional imaging for the detection of mediastinal and internal mammary nodal metastases (sensitivity 85% versus 54% for CT) [15]. In patients with LABC, up to 25% can demonstrate FDG uptake in internal mammary nodes [38]. The identification of unsuspected metastases in the mediastinal and internal mammary nodal regions has a significant impact on patient management and long term prognosis [15]. Patients with positive internal mammary nodes have a significantly worse prognosis for both disease-free and overall survival [15]. Management of these patients may require extending the radiation field to include these nodes (which can result in survival benefit) or initiating a more aggressive chemotherapeutic regimen [15].
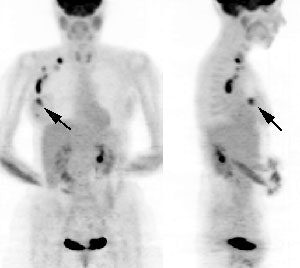
Skeletal metastases are the most common site of initial distant metastatic involvement in patients with breast cancer and account for up to 90% of all metastatic lesions [38,88]. Bone represents the first site of metastases for 26-50% of patients [47,50]. Bone metastases will develop in 30-85% of patients with metastatic breast cancer during the course of disease [47] and can be the sole site for metatases in 28-44% of patients [88]. Breast cancer preferentially metastasizes to the spine and pelvis, followed by the ribs, skull, and femurs [47]. In patients with advanced breast cancer, there is a wide variation in survival rates and about 20% of patients with bone metastases survive more than 5 years [41]. Most metastatic bone lesions from breast cancer are typically "mixed" containing lytic and blastic components [38]. Tc-MDP bone scan has reported sensitivities from 62-100% and specificities from 78-100%- although the exam does suffer from a high false-positive rate [47]. PET imaging has been shown to be superior to bone scan for the detection of osteolytic bone metastases [25], but has a low detection rate for purely osteoblastic metastases [37]. In clinical practice, bone scan and CT remain the standard imaging combination for staging breast cancer, with PET providing complementary information [38]. However, 18F-NaF imaging appears to hold promise for imaging skeletal metastases in breast cancer patients and the results of 18F-NaF imaging can result in a change in patient management planin up to 39% of cases [88]. As with MDP imaging, a flare phenomena can be observed on 18F-NaF imaging both after chemotherapy and endocrine therapy [88].
|
Breast cancer with sclerotic bone mets: The patient below was found to have extensive sclerotic bone metastases on bone scan (left image) and CT (white arrow). Note the lack of tracer uptake on the patient's PET scan (right images). PET is less sensitive for the detection of sclerotic bone metastases, but is superior to bone scan for the detection of lytic lesions. |
|
|
The liver is a common site of metastatic disease from breast cancer [60]. Liver metastases are more common in patients with HER2-overexpressing tumors and these patients also have a higher cummulative incidence of CNS metastases [97]. The median survival of patients with liver metastases is about 18 months [60]. Radioembolization can be used to treat liver metastases in patients with breast cancer with response rates from 39-61% [60]. The change in SUVmax on PET imaging has been shown to be the best predictor of patient survival following radioembolization therapy of hepatic metastases [60].
Although both invasive ductal (IDC) and invasive lobular carcinoma (ILC) commonly metastasize to lymph nodes, bone, and liver, ILCdemonstrates a predeliction for metastases to the peritoneum, retroperitoneum, hollow viscera (including the GI and GU tract), and leptomeninges [84].
In one study, it was suggested that FDG PET/CT is more likely to reveal unsuspected distant metastases in stage III IDC patients than in patients with ILC [84]. In up to 25% of ILC patients upstaged on PET imaging, the lesions were PET negative and identified on the CT images only (sclerotic bone mets) [84].
Monitoring response to therapy:
Systemic therapy is given in the following clinical settings- adjuvant (after completion of definitive locoregional surgery to treat residual or metastatic microscopic disease; neoadjuvant (primary systemic treatment given prior to definitive local surgery); and for metastatic Stage IV disease [48]. The is accumulating clinical evidence that propranolol can reduce the progression of cancer stage, metastasis development, and tumor recurrence in breast cancer patients and improve their disease-free interval and relapse-free survival [75]. Treatment with propranolol can result in a 57% reduced risk of metastasis and a 71% reduction in breast cancer mortality after 10 years [75]. The mechanism of action appears to be related to down-regulation of hexokinase-2 expression (through beta-adenoreceptors) with decreased tumor glucose utilization [75].
|
FDG PET exam results in breast cancer patient guide therapy: The patient shown below was a 40 year old female who had undergone lumpectomy and adjuvant chemo/radiation therapy for breast cancer. Ten months later the patient began to experience pain in the right shoulder. A bone scan and CT scan were interpreted as negative. The FDG PET exam revealed numerous foci of tracer accumulation within the right axilla and chest consistent with metastatic disease. As a result of the PET exam findings the patient was treated with another course of chemo/radiation therapy. The exam was performed on a Siemens ECAT EXACT PET scanner; 10 mCi FDG, and 5 bed positions. Case courtesy of Rush-Presbyterian-St. Luke's Medical Center, Chicago, IL and CTI. |
|
|
Neoadjuvant therapy: Fifteen to 25% of breast cancers are large (over 3 cm) or locally advanced breast cancer (a tumor that is larger than 5 cm (T3 or greater), invades the chest wall or skin, advanced axillary nodal disease (N2), has fixed lymph node metastases, or is inflammatory [27,29]) at the time of initial presentation [5]. Patients with such tumors pose major therapeutic problems due to a high incidence of locoregional recurrence after surgery and an overall poor prognosis [5]. Neoadjuvant (primary chemotherapy) is now increasingly used in the management of these patients in an attempt to "downstage" the primary tumor and eliminate occult metastatic disease [5,25]. Neoadjuvant therapy reduces the recurrence risk, particularly in HER2-positive and triple-negative breast cancers [122]. The rates of pathologic complete response (pCR) to neoadjuvant therapy differ according to tumor subtype [108]. Estrogen receptor negativity, low T-stage, high histoloic grade, and high Ki-67 proliferation have been associated with pCR [108].
Patients with no or only minimal residual disease following completion of primary chemotherapy (pathologic complete response or pCR) have significantly higher disease free and overall survival rates compared to patients with gross residual disease (a complete pathologic response after completion of neoadjuvant therapy occurs in 13-26% of patients [66]) [6,82]. More importantly, studies have demonstrated that patients with unresponsive tumors may achieve an improved survival with the use of alternative and/or more prolonged courses of chemotherapy [5]. It would be of obvious benefit if patients with unresponsive tumors could be identified earlier, thereby avoiding ineffective treatment [5]. Unfortunately, clinical assessment alone cannot accurately predict which patients have responded to therapy and which patients have not [6].
FDG PET imaging offers an alternative method to visualize the effectiveness of induction therapy for various types of breast cancer [6,24,25,32,79]. Changes in tumor glucose uptake (reflected by FDG) can be used to demonstrate and guide effective treatment of breast cancer [5,6] and is more accurate than conventional imaging [32]. Decreased tracer accumulation within a lesion after chemotherapy indicates effective treatment [5,6]. Assessment of therapy response can be determined earlier with FDG PET than with any other method of conventional therapy evaluation (i.e.: before any appreciable decrease in tumor mass can be detected) [2,5,6,24].
To evaluate tumor response to neoadjuvant therapy, imaging is typically performed following two cycles of therapy [93]. Change in SUV has been shown to be statistically higher in patients with pathologic complete response than in those without a complete response [93]. Various optimal thresholds have been applied to assess tumor response and there are no present standard guidelines [93]. The variability can be explained by various definitions of what histopathologically defines a responder, the timing at which the PET scan is performed during therapy, and the variability of breast cancer itself- with multiple tumor histologies, various receptors expressions, response rates to chemotherapy, and risk of relapse [66]. Most studies have shown that the greater the decrease in SUV, the more likely the patient will achieve a complete remission [44]. In a study of patients with triple negative breast cancer, a less than 42% decrease in FDG uptake after 2 cycles of chemotherapy was associated with residual tumor at completion of neoadjuvant treatment and a high risk for early relapse [58]. In patients with bone metastases, a decrease in SUV of 8.5% or more has been shown to be associated with an improved response duration [41]. In general, scans performed mid therapy that demonstrate a decline in FDG uptake by approximately 40-50% are more predictive of a good response, while more modest declines are more characteristic of nonresponders [38,48,91]. A resent meta-analysis found that the best cut-off for a response after two cycles of chemotherapy was a decrease in FDG uptake ranging from 55-65% (NPV 91%, but sensitivity 84% and specificity 66%, are more limited) [89,93].
In the evaluation of response to therapy, PET exam findings can alter management in up to 33% of patients [28].
Although generally performed mid cycle or after two courses of therapy, PET imaging may be able to differentiate responders from non-responders as early as following one cycle of chemotherapy [24]. In two studies, FDG PET imaging after a single pulse of chemotherapy was able to predict a complete pathologic response with a sensitivity of 90-100% and a specificity of 74-85% [5,6]. Smith et al used a decrease in tumor glucose uptake ratio of 20% from the pre-treatment value to identify responders; Schelling et al used a decrease in the tumor glucose uptake ratio below 55% of the baseline scan [5,6].
Because of the lack of a well defined SUV change to be associated with complete pathologic response, other markers of tumor responsiveness have been developed and may be more accurate than SUVmax [68]. The metabolically active tumor volume represents the total volume of tumor as defined by a specified voxel intensity [68]. Total lesion glycolysis represents the product of the MATV and its associated SUV mean [68]. The change in total lesion glycolosis can be a better predictor of tumor response to neoadjuvant therapy with better sensitivity and similar specificity to change in SUV max [68]. The residual metabolic tumor volume has also been shown to be more predictive of risk for recurrence [78].
One limitation of FDG PET imaging is that it is not capable of differentiating patients with residual microscopic disease from those with a complete response [6]. In other words, while residual FDG uptake is predictive of residual disease, the absence of FDG uptake is not a fully reliable indicator of a complete pathologic response [38,48]. None-the-less, FDG PET offers great promise in the early assessment of tumor response to therapy [6].
|
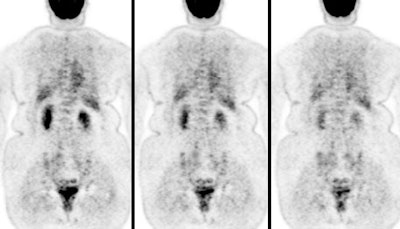
Monitoring response to therapy: The patient shown below had received chemotherapy for treatment of metastatic breast cancer (pre-therapy scan is on the left). The post-therapy scan (right image) demonstrated a very good response to treatment, however, several foci of tracer uptake remained (black arrows) consistent with residual metastatic disease. Further therapy was planned as a result of the PET scan. |
|
|
HER2-positive cancer therapy: The standard of care for patients with stage II-III HER2-positive breast cancer is neoadjuvant chemotherapy in combination with HER2-directed therapy with trastuzumab and pertuzumab [121]. In the PHERGAIN trial, patients were treated with 2 cycles of HER2-directed therapy with trastuzumab and pertuzumab and then randominzed to receive either standard chemotherapy with HER2-directed treatment or HER2-directed therapy alone, based on the results of their interim PET scan results [121]. Patients that achieved a 40% or greater decline in SUVmax at week 6 were continued on HER2-directed therapy without chemotherapy [121]. This patient cohort achieved a pCR of almost 40% and had only one documented local relapse at 3 year follow-up [121].
Prognosis:
Triple-negative breast cancer accounts for 15% of invasive breast cancers and is characterized by the lack of estrogen and progresterone receptors and the absence of HER2 overexpression [58]. Patients with triple negative breast cancer have a relatively poor outcome with a higher relapse rate [58]. However, patients that have a complete pathologic response to neoadjuvant therapy have a good prognosis with a 3 year overall survival of 94% (compared to 68% for patients with residual tumor at the time of surgery) [58].
For patients with locally advanced breast cancer high uptake on FDG imaging may predict a poor response to neoadjuvant chemotherapy [18,32]. High SUV max has also been found to correlate with higher nuclear grade and triple negative hormone receptor profile (a profile associated with aggresive biologic behavior) [76].
For patients with distant metastatic disease at presentation, whole body metabolic tumor volume (MTV) has been shown to be an independent prognostic factor for overall survival and superior to primary tumor and nodal mets SUVmax [80].
FDG PET imaging is superior to conventional imaging in predicting outcome following initiation of therapy in breast cancer patients [14,32]. A negative post-treatment FDG PET scan is a powerful predictor of survival and superior to CT imaging [89]. Residual tumor metabolic activity after completion of therapy is an indicator of residual viable tumor tissue [89]. Patients with post-therapy positive FDG PET exams have a significantly shorter disease free survival when compared to patients with negative PET studies [14,32]. FDG PET imaging is particularly useful in post-therapy cases in which discordant results are found when compared to conventional imaging [14]. In these cases, PET imaging has been shown to be correct up to 4 times more frequently than conventional imaging [14].
Reports suggest that incidental diffuse thyroid uptake on PET imaging related to autoimmune thyroiditis in patients with advanced breast cancer (either preoperatively or postoperatively) is a favorable prognostic indicator independently associated with low tumor recurrence [64].
Evaluation of tumor recurrence:
Approximately 30% of patients with early-stage breast cancer develop recurrent disease, and recurrence continues to occur up to 15 years after definitive treatment has been received [90]. The annual hazard of recurrence peaks in the second year following diagnosis, but the annual risk remains 2-5% in years 5 to 20 [106]. The clinical course of patients with recurrent breast cancer varies and is largely dependent on the extent of metastatic disease and the biological aggressiveness of the tumor [12]. Accurate staging is critical for therapeutic planning [12]. Unfortunately, previous surgery and radiation therapy can result in scarring that can complicate the evaluation for locoregional recurrence of breast cancer- particularly in the axillary region [12,13]. Exploratory surgery of the axilla has been used to confirm or exclude recurrent disease, but can yield inconclusive results [13] due to sampling error. Furthermore, early distant metastatic disease may not be suspected clinically or may not be appreciated on conventional imaging.
FDG PET has demonstrated usefulness in discriminating tumor recurrence from post-operative and post-radiation changes in the breast and PET imaging has also been shown to add incremental information to conventional imaging studies in defining disease extent [12,13,28]. In one study comparing FDG PET vs CT in monitoring for breast cancer recurrence, recurrence was first detected on PET imaging in 49% of patients, compared to only 1% on CT, and on average 6 months sooner than on CT iamging [117]. Earlier detection of tumor recurrence offers the opportunity for earlier treatment which may improve overall survival [117]. FDG PET imaging is recommended in addition to conventional imaging for the detection of recurrent breast cancer [40] and patient management can be modified in 48-57% of patients with suspected relapse following PET/CT imaging [86]. SUVmax, TLG (SUVmean x MTV), and total tumor volume have been shown to provide integrated risk stratification and prognostic survival information in patients with recurrent breast cancer [90]. Patients with both high SUVmax (2.9 or greater) and high MTV (7.95 mL or more) have signficantly poorer survival [90].
Locoregional recurrence: Local or regional recurrence of breast cancer occurs after initial diagnosis and treatment in 7-30% of patients [13]. After conservative surgical resection and radiation, the most common sites of locoregional recurrence are the breast, the skin of the breast, chest wall, and local nodal basins [12]. In these patients, risk factors for locoregional recurrence include positive margins at surgery, age under 40 years, and tumors containing an extensive intraductal component [12]. Following mastectomy, axillary node dissection, and adjuvant chemotherapy, the most common sites of locoregional recurrence are the chest wall and supraclavicular nodes [12]. In these cases, risk factors for local recurrence include four or more positive axillary nodes, a tumor diameter over 4 cm, and extranodal extension of greater than 2 mm [12]. These patients have a poor prognosis in terms of survival following recurrence [12].
FDG PET has a reported sensitivity of up to 100% for the detection of locoregional recurrence [13]. For patients with suspected or known locoregional recurrence, under consideration for aggressive local treatment, PET imaging findings can significantly affect patient management [28]. By detecting unsuspected sites of disease, planned therapy can be altered in up to 44% of patients [28]. In one study of patients with elevated tumor markers, PET/CT was 90% sensitive for diagnosing recurrent tumor and affected clinical management in 51% of patients [38].
|
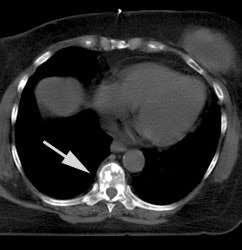
FDG PET exam for the evaluation of locoregional recurrence: The patient shown in the case below had a history of breast cancer and had developed left chest pain. She presented for the evaluation of possible metastatic disease. The CT scan revealed extensive soft tissue thickening in the left breast which was felt possibly related to scar from prior surgery and radiation therapy. There was a 2 cm lymph node in the left axilla (not shown) which was concerning for metastatic disease. Axial (center) and coronal (right) images from the patients FDG PET exam demonstrated marked increased FDG accumulation within the left breast corresponding to the soft tissue abnormality on CT. There were also multiple foci of increased uptake within the the left axilla. Biopsy revealed recurrent breast cancer. Case courtesy of CTI, The power behind PET. |
|
|
Distant recurrence: Internal mammary (IM) and mediastinal nodes represent sites of intrathoracic recurrence. FDG PET imaging has been shown to be superior to CT for the detection of IM nodes [12,15]. Note that nonmalignant IM node enlargement (up to 38% of patients) and FDG avidity (up to 21% of patients referred for PET) can be seen following reconstructive oncoplastic surgery using silicone implants in breast cancer patients [83].
Bone is the most common site of breast cancer metastases and the most frequent site for recurrence after treatment (the first site of relapse is the bone in half of breast cancer patients [106]) [30]. Bone metastases affect 8% of all patients who develop breast cancer, but this increases to 70% in patients with advanced disease [16]. FDG is more sensitive for the detection of lytic lesions or those predominantly involving the bone marrow, while bone scan is much more sensitive for osteoblastic lesions [12,16,30]. This likely related to a higher glycolytic rate in lytic metastases compared with osteoblastic lesions [16]. Overall- bone scanning with SPECT has been suggested to be superior to PET for the detection of breast cancer bone metastases due to an overall higher percentage of osteoblastic lesions in breast cancer patients [31]. However, patients with purely lytic bone lesions have a lower survival compared with patients with sclerotic or mixed metastases and may benefit from more aggressive or newer therapies [16]. Therefore, FDG PET imaging is not a substitute for bone scan in patients with breast cancer- however, the two modalities are complementary [12,16,30,31]. 18F-Fluoride PET has also been evaluated in patients with breast cancer and the agent is able to detect small lesions with minimal bone reaction [33]. Findings on the 18F-Fluoride PET scan can result in change in management in almost 12% of patients [33].
|
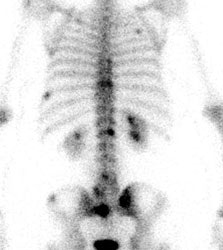
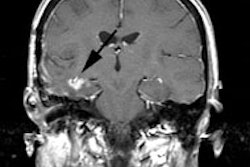
Unsuspected asymptomatic bone metastases: The patient shown below had a history of left breast cancer treated with mastectomy and adjuvant chemo/radiation therapy. The patient presented with a new palpable fullness in the left infraclavicular region. A CT exam revealed a large mass consistent with recurrent tumor (white arrow) and PET imaging was performed for further staging. In addition to uptake in the supraclavicular region (black arrow), PET imaging demonstrated unsuspected, asymptomatic bone metastases to the sacrum, lumbar spine, and left ischium (blue arrows) which were confirmed on MRI (sacrum ischium). |
|
|
Dedicated mammographic PET:
Dedicated high-spatial resolution mammographic PET imaging units have been developed- positron emission mammography or PEM - and have been shown to be superior to whole-body PET systems for the evaluaiton of breast lesions [52,53,74]. These units can provide full tomographic image reconstruction with a spatial resolution of 1.6 mm [74]. Two types of systems are available- positron emission mammography (PEM- which employs light compression of the breasts between two opposed plate-like detectors) and a tomographic technique with a ring-shaped scanner [101].
Pooled data from PEN studies for the evaluation of suspected breast lesions have demonstrated a sensitivity of 87% and a specificity of 85% [74]. Sensitivities for subcentimeter cancers and DCIS have been reported to be 63% and 88%, respectively [74]. These units have been shown to have a high positive predictive value (0.88) and overall high sensitivity (90%) for the detection of malignancy not seen on mammogram or breast ultrasound [52]. In women with breast cancer, up to 15% of malignancies may be seen only on PEM [53]. Dedicate PET mammography has been shown to be complementary to breast MR for defining the pre-operative extent of tumor in women with newly diagnosed breast cancer [52].However, other studies suggest that MR is more sensitive than PEM- particularly in patients with dense breasts [56]. In that study, PEM detected only 20% of contralateral breast cancers prospectively and 21% of lesions classified as "probably benign" proved to be malignant [56]. However, tracer uptake in a lesion should be viewed as suspicious for malignancy [56].
Other PET applications in breast cancer:
18F-fluoroestradiol (FES) imaging:
Approximately 80% of breast cancers are hormone receptor positive - either estrogen receptor (ER) and/or progesterone receptor (PR) positive [118]. Nearly two-thirds of invasive breast carcinomas express the estrogen receptor (ER) [107]. Patients with HR-positive disease can be treated with hormone therapy including aromatase inhibitors (anastrozole), selective ER modulators (tamoxifen), and selective ER down-regulators (fulvestrant) [118]. The ER status of breast cancers has been shown to carry important prognostic information and will greatly influence the choice of systemic therapy [22,39]. In general, hormone-sensitive breast cancer occurs more commonly in postmenopausal women, is less aggressive than hormone resistance disease, and is characterized by a more favorable prognosis with longer disease free and overall survival [59,92]. Patients with localized HR-positive disease have a 5-year survival rate of 99-100% compared to 91-97% for patients with HR-negative localized breast cancer [118]. Patients with metastatic HR-positive breast cancer have a 5-year survival of 32-46%, compared to 12-40% for patients with HR-negative metastatic disease [118].
Absence of ER expression has a strong negative predictive value for response to antihormonal therapy [57]. About 70-75% of breast cancers are estrogen receptor positive (ER+) [55,92]. Absence of ER receptors indicates a low likelihood of response to ER-targeted therapy [92]. However, not all (ER+) patients will respond to ER-directed adjuvant therapy [55]. Only between 30% to 60% of patients with estrogen-receptor positive (ER+) advanced breast cancer will objectively respond to antiestrogen therapy (tamoxifen) and a substantial faction of patients will have stable disease for a clinically significant period [22,57]. Additionally, there can be discordant ER expression between the primary tumor and metastatic lesions in 18-55% of patients (i.e.: loss of ER expression in sites of metastatic disease) [57,100]. In one study, 36% of patients had both FES positive and negative lesions confirming heterogeneous disease [100]. Additionally, the ER expression of tumors may change over time [109]. The presence of progesterone receptor (PR +) increases the likelihood of responsiveness to antiestrogen therapy to approximately 75% [59]. PR-negative tumors are less responsive to hormone therapy [59]. Only a small fraction of tumors are ER- and PR+, and these lesions demonstrate an intermediate response to endocrine therapy [59].
In endocrine resistant tumors, histone deacetylase inhibitors (HDACIs) may reverse endocrine resistance in ER+ metastatic breast cancers [107].
ER expression may vary spatially and temporally, and the results obtained from tissue sampling may incompletely represent a patient's ER receptor distribution [116]. Moreover, not all tumors that are ER-positive by immunohistochemistry respond to ER-targeted therapy [116]. The estrogen receptor status of breast cancers can be reliably evaluated with PET using the radiolabeled estrogen analog F-18 estradiol (FES) [109]. 18F-FES PET can demonstrate ER heterogeneity within individual patients, serves as a prognostic biomarker, provides high diagnostic accuracy for the detection of ER-positive metastases, and can assess the efficacy of ER blackade [116].
Using biopsy as the gold standard, 18F-FES PET has been shown to have a sensitivity of 98% and a specificity of 78% for assessment of ER status [109]. Uptake of the tracer in tumor lesions correlates well with ER expression measured with immunohistochemistry [100]. The agent has been approved for use in patients with ER-positive recurrent or metastatic breast cancer as an adjunct to biopsy [118].
Patient preparation:
Fasting is not required prior to imaging and imaging can be performed on pre-menopausal patients because physiologic estrogen levels do not affect FES uptake [118]. Lactating women are recommended to avoid breast feeding for 4 hours after FES administration [118].
FES imaging can be performed on patients that are taking aromatase inhibitors which decrease peripheral and local estrogen production, but do not block estrogen receptors [118]. FES can also be performed in patients taking cyclin-dependent kinase (CDK4 or CDK6) inhibitors, because they also do not interact with the estrogen receptors [118].
It is highly recommended that patients be imaged before they start ER-antagonist therapy [118]. ER blockade with ER-antagonist therapies decreases or completely blocks FES binding to the ER [118].
Because tamoxifen can block tumor FES uptake, when evaluating for the ER receptor status of a tumor, it should ideally be discontinued for 5 weeks prior to imaging (other authors indicate that tamoxifen should be held for 8 weeks prior imaging) and that fulvestrant - with a half-life of 40 days- should be held for 28 weeks) [57,118]. The SUVmax of metastases has been shown to be 42% lower in patients that received ER antagonists before FES PET imaging (stopping antagonists 3-12 weeks before imaging) [100]. Also- the percentage of FES negative lesions has been shown to be higher in patients that recently stopped ER antagonist use with an absolute difference of 25% [100]. This may be related to competition for ER receptors, downregulation of ER, or selection of ER negative clones in patients treated with ER antagonists [100].
Distribution:
18F-FES binds to the estrogen receptor in the nucleus of ER-expressing cells [118]. FES activity can be normally seen in the liver (the critical organ), gallbladder/biliary system, small bowel,kidneys, ureters, urinary bladder , and uterus [118]. The normal endometrium has high ER expression and demonstrates FES uptake in both pre- and postmenopausal patients [118]. Confirmation of uterine uptake can serve as an internal control supporting adequate FES tissue targeting [118]. The vein draining from the injection site often contains residual radiotracer [118]. High physiologic liver activity limits evaluation for liver metastatic disease [118]. Tracer uptake can be seen in uterine fibroids and meningiomas [118].
18F-FES is highly extracted and metabolized by the liver, resulting in rapid early blood clearance [92]. By 20 minutes after injection, only 20% of the total activity is attributable to unmetabolized FES [92]. Unmeabolized FES is highly protein bound in blood [92]. The agent has a much higher binding affinity for sex-hormone binding globulin (SHBG) than for albumin, but the higher concentration of albumin in the blood results in an approximately 1:1 distribution of FES between SHBG and albimun [92]. The non-bound metabolites of the agent (comprised of glucuronide and sulfate conjugated products) are secreted in the bile, resorbed via the enterohepatic circulation, and renally excreted [92]. Efficient resorption from the small bowel back into the bloodstream leads to minimal to no distal large bowel activity [118].
Unlike FDG, FES uptake is typically not seen in inflammatory processes, degenerative change, or reactive bone marrow [118]. However, an exception is radiation pneumonitis (possibly related to extravasation from leaky vessels) and in atelectasis [118].
Exam:
The recommend dose of FES is 6 mCi (222 MBq) and it is preferably given through a syringe pump over a 1-2 minute infusion followed by a flush of at least 10 mL of normal saline [118]. Imaging is optimally performed 60-80 minutes after injection [118]. Patients should void immediately before imaging to decrease urinary bladder activity.
At the highest recommended dose (2.22 x 108 Bq or 6 mCi) the effective dose equivalent is 0.002 mSv/MBq, with the critical organ being the liver (dose 0.13 mSv/MBq) [92]. FES uptake has been shown to be inversely associated with plasma SHBG levels (protein bound FES may be less available to tissue ER receptors and this results in decraesed FES uptake) [92]. For the detection of ER+ breast cancer, FES imaging has a sensitivity of 84% and a specificity of 98% [95]. 18F-FES has a 6.3 fold absolute affinity preference for ER-alpha over ER-beta [115].
A major advantage of FES PET imaging is the ability to assess the ER status of tumor lesions across the whole body [92]. FES studies have demonstrated FES-negative disease in 32-53% of ER+ patients [57]. Heterogeneous FES uptake can also be seen with some patients demonstrating both FES-positive and negative lesions, suggesting loss of ER expression in some lesions [92]. The level of FES uptake can predict response to tamoxifen therapy in locally advanced and metastatic breast cancer, while low FES uptake is a strong predictor of failure of antihormonal therapy [57].
The SUV of FES positive diease is generally lower than that seen with other radiotracers [118]. A tumor with a SUV value of greater than 1.0 on FES imaging is considered ER positive [22], however, other authors indicate an SUVmax of 1.5 or greater to represent FES-positive disease [118]. Sites of abnormal uptake can also be identified qualitatively showing higher than local and blood pool background uptake [118]. Some studies have suggested that tumors with an SUVmax below 1.5 are unlikely to respond to endocrine therapy [95] (other authors suggest an SUVmax of less than 7.3 is associated with lack of response to neoadjuvant endocrine therapy [96]). In an analysis of four studies, 88% of patients with tumor FES uptake of less than 1.5 who underwent endocrine therapy experienced treatment failure (NPV 88% and the PPV of treatment response was 65%) [118]. Patients with heterogeneous FES uptake (both FES positive and negative metastases) have also been shown to have worse outcomes [118].
Patients with discordant in vitro ER expression and FES results (ER positive, but FES negative) tend to have a decreased response to hormone therapy, suggesting the lesions are functionally hormone therapy-resistant [92]. ER-rich tumors with poor FES uptake may be better treated with neoadjuvant chemotherapy [95,96].
Because FES accumulation is a receptor mediated process that can be blocked with anti-estrogen therapy- treatment response can be monitored as soon as 7 to 10 days following initiation of therapy [1,22]. Patients with incomplete ER blockade at FES PET have been shown to have earlier disease progression than those with complete ER bloackade [118]. The degree of ER blockade can be measured by a decrease in SUV and responders have be shown to have a greater decrease in SUV (2.7 +/- 1.7) than non-responders (0.8 +/- 0.5) [22] (mean percentage decrease in FES uptake 55% +/- 14% for responders versus 19% +/- 17% for non-responders [92]). Another study demonstrated that a less than 75% decrease in median tumor SUV was significantly associated with shorter progression-free survival [92]. Patients that respond to tamoxifen often have a higher baseline tumor SUV's compared to non-responders [22].
The estrogen receptor status of a patient's tumor can also provide insight into potential sites of metastatic disease. The liver is the most common visceral organ for breast cancer metastases and patients with estrogen-receptor negative tumors have an increased risk for liver metastases [12]. Unfortunately, the liver and lesions directly adjacent to the liver are not well evaluated with FES PET because of tracer metabolism in the liver that results in high tracer accumulation [55,57]. Because of high liver activity and discordant loss of ER receptors in some metastatci foci, FES imaging cannot be used to reliably exclude metastatic disease [57]. Up to 45% of patients have been shown to have concurrent FES-positive and FES-negative metastases [57].
Indirect evidence of effective hormone therapy can be assessed with PET FDG imaging to look for a "metabolic flare". A symptomatic "clinical flare" occurs in a small number of patients following initiation of tamoxifen therapy and can be difficult to distinguish from disease progression [22]. The condition is felt to be related to agonist effects of antiestrogen therapy which occur within 7 to 10 days of initiation of treatment (before the antagonist effects take precedence) [34,66]. Patients experience increased pain at sites of osseous metastatic disease, erythema in soft tissue lesions, hypercalcemia, and apparent disease progression on bone scintigraphy [23]. The development of a clinical hormone flare reaction is usually predictive of a positive response to therapy with disease remission with subsequent response in 80% of patients [22]. Although the etiology of the clinical flare is not definitively known, it may be related to temporary agonist effects of tamoxifen on the tumor [22]. By using FDG imaging, a metabolic tumor flare can be identified in patients 7 to 10 days following initiation of tamoxifen therapy and is characterized by an increase in tumor SUV (1.4 +/- 0.7) [44]. The presence of a metabolic flare is a strong indicator of subsequent response to therapy [22,51,66,92]. The lack of a metabolic flare is indicative a non-response to the therapy [22]. The flare phenomenon has not been described following chemotherapy [26]. A subsequent healing osteoblastic flare can be identified on 18F-fluoride imaging and there is corresponding decreased FDG uptake [35].
Other breat imaging tracers:
Human epidermal growth factor receptor type 2 (HER 2, ErbB2, neu) is a membrane tyrosine kinase-linked growth factor receptor- unchecked hyperactivity of the receptor (due to HER 2 overespression) upregulates cell proliferation in breast cancer and poor clinical outcome [46,49,112]. HER2 promotes cell proliferation, angiogenesis, resistance to apoptosis, and tumor metastases [71,72]. About 20% of breast cancer patients have high expression of HER2 and these cancers may respond to anti-HER 2 antibody treatment [46,71,73]. Trastuzumab is a humanized monoclonal antibody therapy targeting HER2 and when combined with chemotherapy the agent has produced dramatic improvements in recurrence rates and overall survival in patients with HER2 overexpressing tumors [71,73]. Lapatinib is another agent that has a dual mode of action - targeting both HER2 as well as the epidermal growth factor receptor [71]. However, not all patients with HER 2 overexpression demonstrate a response to therapy [73]. This is likely due to differences in HER2 expression between the primary and metastatic lesions which has been observed in 20% of patients (particularly when metastases occur after adjuvant or neoadjuvant therapy [73].
68Ga-labeled (Fab'2) trastuzumab (Herceptin) and 64Cu-DOTA-trastuzumab are radiolabeled biomakers for the expression of HER 2 growth factor receptors [46,73].
FDG PET imaging can be used in HER2 overexpressing patients to determine early response to therapy [71]. As early as 2 weeks following initiation of HER2 therapy, responders demonstrate a significantly larger decrease in SUVmax compared to non-responders [71,72].
Tumor hypoxia:
FMISO binds intracellularly under hypoxic conditions (it does not bind to necrotic cells) and it can be used to evaluate tumor hypoxia [67]. Approximately 70% of breast cancers over-express hormone receptors [67]. As a result, endocrine therapy has emerged as an important ool for the treatment of ER-positive breast cancers [67]. However, between 30-40% of ER-positive tumors display primary resistance to hormonal therapy [67]. Tumor hypoxia may play a role in this endocrine therapy resistance [67].
Fibroblast activation protein inhibitors (FAPI):
The fibroblast activation protein is a stromal antigen expressed by activated fibroblasts, including cancer associated fibroblasts, whereas expression in healthy tissue is virtually absent [110]. 68Ga-FAPI has been shown to accumulate in primary breast lesions and lymph node metastases [110]. In contrast to FDG, no association of tracer uptake with grade, receptor status, or histologic type (lobular or ductal) was noted with 68Ga-FAPI [110,111].
REFERENCES: (1) J Nucl Med 1995; Dehdashti F, et al. Positron tomographic assessment of estrogen receptors in breast cancer: Comparison with FDG-PET and in-vitro receptor assays. 36 (10): 1766-74
(2) J Clin Onc 2000; Avril N, et al. Breast imaging with positron emission tomography and fluorine-18 fluorodeoxyglucose: Use and limitations. 18: 3495-3502
(3) Radiologic Clinics of North America 2000; Pisano ED, et al. Digital mammography, sestamibi breast scintigraphy, and positron emission tomography breast imaging. 38: 861-869
(4) J Comput Assist Tomogr 2000; Yutani K, et al. Comparison of FDG-PET with MIBI-SPECT in the detection of breast cancer and axillary lymph node metastasis. 24: 274-280
(5) J Clin Oncology 2000; Smith IC, et al. Positron emission tomography using [18F]-fluorodeoxy-D-glucose to predict the pathologic response of breast cancer to primary chemotherapy. 18: 1676-1688
(6) J Clin Oncology 2000; Schelling M, et al. Positron emission tomography using [18F] fluorodeoxyglucose for monitoring primary chemotherapy in breast cancer. 18: 1689-1695
(7) J Nucl Med 2001; Avril N, et al. Glucose metabolism of breast cancer assessed by 18F-FDG PET: Histologic and immunohistochemical tissue analysis. 42: 9-16
(8) J Nucl Med 2001; Murthy K, et al. Results of preliminary clinical trials of positron emission mammography system PEM-I: A dedicated breast imaging system producing glucose metabolic images using FDG. 41: 1851-1858
(9) J Nucl Med 2001; van Rijk PP, van Dongen AJ. Limited angle, limited approach? 41: 1859-1860 (No abstract available)
(10) J Cancer Res Clin Oncol 2000; Ilknur AK, et al. Positron emission tomography with 2-[18F] fluoro-2-deoxy-D-glucose in oncology: Part II: The clinical value in detecting and staging primary tumours. 126: 560-574 (No abstract available)
(11) J Nucl Med 2001; Yap CS, et al. Impact of whole-body 18F-FDG PET on staging and managing patients with breast cancer: The referring physicians perspective. 42: 1334-1337
(12) Radiographics 2002; Eubank WB, et al. Detection of locoregional and distant recurrences in breast cancer patients using FDG PET. 22: 5-17
(13) Radiology 1999; Hathaway PB, et al. Value of FDG PET and MR imaging in the evaluation of suspected recurrent local-regional breast cancer: preliminary experience. 210: 807-14
(14) J Nucl Med 2002; Vranjesevic D, et al. Whole-body 18FDG PET and conventional imaging for predicting outcome in previously treated breast cancer patients. 43: 325-329
(15) J Clin Oncol 2001; Eubank WB, et al. 18Fluorodeoxyglucose positron emission tomography to detect mediastinal or internal mammary metastases in breast cancer. 19: 3516-3523
(16) J Clin Oncol 1998; Cook GJ, et al. Detection of bone metastases in breast cancer by 18FDG PET: differing metabolic activity in osteoblastic and osteolytic lesions. 16: 3375-3379
(17) J Clin Oncol 2002; Bos R, et al. Biologic correlates of 18Fluorodeoxyglucose uptake in human breast cancer measured by positron emission tomography. 20: 379-387
(18) J Nucl Med 2002; Mankoff DA, et al. Blood flow and metabolism in locally advanced breast cancer: relationship to response to therapy. 43: 500-509
(19) Eur J Nucl Med 2001; Schirrmeister H, et al. Fluorine-18 2-deoxy-2-fluoro-D-glucose PET in the preoperative staging of breast cancer: comparison with the standard staging procedures. 28: 351-8
(20) J Natl Cancer Inst 2001; Greco M, et al. Axillary lymph node staging in breast cancer by 2-fluoro-2-deoxy-D-glucose-positron emission tomography: clinical evaluation and alternative management. 93: 630-5
(21) Clin Positron Imaging 2000; Hubner KF, et al. The Potential of F-18-FDG PET in Breast Cancer. Detection of Primary Lesions, Axillary Lymph Node Metastases, or Distant Metastases. 3: 197-205
(22) Eur J Nucl Med 1999; Dehdashti F, et al. Positron emission tomographic assessment of "metabolic flare" to predict response of metastatic breast cancer to antiestrogen therapy. 26: 51-56
(23) Semin Roentgenology 2002; Dehdashti F, Siegel BA. Evaluation of breast and gynecologic cancers by positron emission tomography. 37: 151-168
(24) J Nucl Med 2003; Kostakoglu L, Goldsmith SJ. 18F-FDG PET evaluation of the response to therapy for lymphoma and for breast, lung, and colorectal carcinoma. 44: 224-239
(25) Radiographics 2003; Kostakoglu L, et al. Clinical role of FDG PET in evaluation of cancer patients. 23: 315-340
(26) J Nucl Med 2003; Biersack HJ, Palmedo H. Locally advanced breast cancer: is PET useful for monitoring primary chemotherapy. 44: 1815-1817 (No abstract available)
(27) J Clin Oncol 2004; van der Hoeven JJ, et al. 18F-2-fluoro-2-deoxy-D-glucose positron emssion tomography in staging of locally advanced breast cancer. 22: 1253-1259
(28) J Nucl Med 2004; Eubank WB, et al. Impact of FDG PET in defining the extent of disease and on the treatment of patients with recurrent or metastatic breast cancer. 183: 479-486
(29) J Nucl Med 2004; Tseng J, et al. 18F-FDG kinetics in locally advanced breast cancer: correlation with tumor blood flow and changes in response to neoadjuvant chemotherapy. 45: 1829-1837
(30) AJR 2005; Uematsu T, et al. Comparison of FDG PET and SPECT for detection of bone metastases in breast cancer. 184: 1266-1273
(31) Radiol Clin N Am 2004; Kumar R, Alavi A. Fluorodeoxyglucose-PET in the management of breast cancer. 42: 1113-1122
(32) J Nucl Med 2005; Early prediction of response to chemotherapy in metastatic breast cancer using sequential 18F-FDG PET. 46: 1144-1150
(33) J Nucl Med 2005; Even-Sapir E. Imaging of malignant bone involvement by morphologic, scintigraphic, and hybrid modalities. 46: 1356-1367
(34) Radiol Clin N Am 2005; Avril NE, Weber WA. Monitoring response to treatment in patients utilizing PET. 43: 189-204
(35) AJR 2006; Flare response in 18F-fluoride ion PET bone scanning. 186: 1783-1786
(36) J Nucl Med 2006; Mavi A, et al. Dual time point 18F-FDG PET imaging detects breast cancer with high sensitivity and correlates well with histologic subtypes. 47: 1440-1446
(37) Radiographics 2007; Lim HS, et al. FDG PET/CT for detection and evaluation of breast diseases: usefulness and limitations. 27: S197-S213
(38) Radiographics 2007; Rosen EL, et al. FDG PET, PET/CT, and breast cancer imaging. 27: S215-S229
(39) J Nucl Med 2008; Peterson LM, et al. Quantitative imaging of estrogen receptor expression in breast cancer with PET and 18F-fluoroestradiol. 49: 367-374
(40) J Nucl Med 2008; Fletcher JW, et al. Recommendations on the use of 18F-FDG PET in oncology. 49: 480-508
(41) Radiology 2008; Tateishi U, et al. Bone metastases in patients with metastatic breast cancer: morphologic and metabolic monitoring of response to systemic therapy with intergrated PET/CT. 247: 189-196
(42) J Nucl Med 2008; Heusner TA, et al. Breast cancer staging in a single session: whole-body PET/CT mammography. 49: 1215-1222
(43) AJR 2008; Imbriaco M, et al. Dual-time-point 18F-FDG PET/CT versus dynamic breast MRI of suspicious breast lesions. 191: 1323-1330
(44) J Nucl Med 2009; Ben-Haim S, Ell P. 18F-FDG PET and PET/CT in the evaluation of cancer treatment response. 50: 88-99
(45) J Nucl Med 2009; Carkaci S, et al. Retrospective study of 18F-FDG PET/CT in the diagnosis of inflammatory breast cancer: preliminary data. 50: 231-238
(46) J Nucl Med 2009; Zhao B, et al. Imaging surrogates of tumor response to therapy: anatomic and functional biomarkers. 50: 239-249
(47) J Nucl Med 2009; Lee JH, et al. The role of radiotracer imaging in the diagnosis and management of patients with breast cancer: part I- overview, detection, and staging. 50: 569-581
(48) J Nucl Med 2009; Lee JH, et al. The role of radiotracer imaging in the diagnosis and management of patients with breast cancer: part 2 - response to therapy, other indications, and future directions. 50: 738-748
(49) J Nucl Med 2009; Kramer-Marek G, et al. Changes in HER2 expression in breast cancer xenografts after therapy can be quantified using PET and 18F-labeled affibody molecules. 50: 1131-1139
(50) J Nucl Med 2010; De Giorgi U, et al. 18F-FDG PET/CT findings and circulating tumor cell counts in the monitoring of systemic therapies for bone metastases from breast cancer. 51: 1213-1218
(51) J Nucl Med 2010; Linden HM, Mankoff DA. Breast cancer and hormonal stimulation: is glycolysis the first sign of response? 51: 1663-1664
(52) Radiology 2011; Berg WA, et al. Breast cancer: comparitive effectiveness of positron emission mammography and MR imaging in presurgical planning for the ipsilateral breast. 258:59-72
(53) AJR 2011; Narayanan D, et al. Interpretation of positron emission mammography: feature analysis and rates of malignancy. 196: 956-970
(54) J Nucl Med 2011; Groheux D, et al. The yield of 18F-FDG PET/CT in patients with clinical stage IIA, IIB, or IIIA breast cancer: a prospective study. 52: 1526-1534
(55) J Nucl Med 2011; Kurland BF, et al. Between-patient and within-patient (site-to-site) variability in estrogen receptor binding, measured in vivo by 18F-fluoroestradiol PET. 52: 1541-1549
(56) AJR 2012; Berg WA, et al. Comparative effectiveness of positron emission mammography and MRI in the contralateral breast of women with newly diagnosed breast cancer. 198: 219-232
(57) J Nucl Med 2012; van Kruchten M, et al. PET imaging of estrogen receptors as a diagnostic tool for breast cancer patients presenting with a clinical dilemma. 53: 182-190
(58) J Nucl Med 2012; Groheux D, et al. Triple-negative breast cancer: early assessment with 18F-FDG PET/CT during neoadjuvant chemotherapy identifies patients who are unlikely to achieve a pathologic complete response and are at high risk of early relapse. 53: 249-254
(59) J Nucl Med 2012; Dehdashti F, et al. Assessment of Progesterone Receptors in Breast Carcinoma by PET with 21-18F-Fluoro-16α,17α-[(R)-(1'-α-furylmethylidene)Dioxy]-19-Norpregn-4-Ene-3,20-Dione. 53: 363-370
(60) J Nucl Med 2012; Haug AR, et al. 18F-FDG PET/CT predicts survival after radioembolization of hepatic metastases from breast cancer. 53: 371-377
(61) AJR 2012; Kizmanich-Conniff KM, et al. Triple receptor-negative breast cancer: imaging and clinical characteristics. 199: 458-464
(62) J Nucl Med 2012; Song BI, et al. 18F-FDG uptake by metastatic axillary lymph nodes on pretreatment PET/CT as a prognostic factor for recurrence in patients with invasive ductal breast cancer. 53: 1337-1344
(63) J Nucl Med 2012; Iima M, et al. Clinical performance of 2 dedicated PET scanners for breast imaging: initial evaluation. 53: 1534-1542
(64) J Nucl Med 2012; Kim SS, et al. Incidental diffuse thyroid 18F-FDG uptake related to autoimmune thyroiditis may be a favorable prognostic factor in advanced breast cancer. 53: 1855-1862
(65) J Nucl Med 2013; Groheux D, et al. 18F-FDG PET/CT in staging patients with locally advanced or inflammatory breast cancer: compasrison to conventional staging. 54: 5-11
(66) Radiology 2013; Groheux D, et al. Performance of FDG PET/CT in the clinical management of breast cancer. 266: 388-405
(67) J Nucl Med 2013; Cheng J, et al. 18F-FDG-fluoromisonidazole PET/CT: a potential tool for predicting primary endocrine therapy resistance in breast cancer. 54: 333-340
(68) J Nucl Med 2013; Hatt M, et al. Comparison between 18F-FDG PET image-derived indices for early prediction of response to neoadjuvant chemotherapy in breast cancer. 54: 341-349
(69) AJR 2013; Wang CL, et al. 18F-FDG PET/CT-positive internal mammary lymph nodes: apthologic correlation by ultrasound-guided fine-needle aspiration nand assessment of associated risk factors. 200: 1138-1144
(70) J Nucl Med 2013; Gemignani ML, et al. Feasibility and predictability of perioperative PET and estrogen receptor ligand in patients with invasive breast cancer. 54: 1697-1702
(71) J Nucl Med 2013; Kenny LM. 18F-FDG PET/CTfor early prediction of response to neoadjuvant lapatinib, trastuzumab, and their combination in HER2-positive breast cancer: results from neo-ALTTO. 54: 1855-56
(72) J Nucl Med 2013; Genhart G, et al. 18F-FDG PET/CTfor early prediction of response to neoadjuvant lapatinib, trastuzumab, and their combination in HER2-positive breast cancer: results from neo-ALTTO. 54: 1862-1868
(73) J Nucl Med 2014; Mortimer JE, et al. Functional imaging of human epidermal growth factor receptor 2-positive metastatic breast cancer using 64Cu-DOTA-trastuzumab PET. 55: 23-29
(74) J Nucl Med 2014; Fowler Am. A molecular approach to breast imaging. 55: 177-180
(75) J Nucl Med 2014; Kang F, et al. Propranolol inhibits glucose metabolism and 18F-FDG uptake of breast cancer through posttranscriptional downregulation of hexokinase-2. 55: 439-445
(76) J Nucl Med 2014; Baba S, et al. Diagnostic and prognostic value of pretreatment SUV in 18F-FDG/PET in breast cancer: comparison with apparent diffusion coefficient from diffusion-weighted MR imaging. 55: 736-742
(77) J Nucl Med 2014; Riedl CC, et al. Retrospective analysis of 18F-FDG/PET/CT for staging asymptomatic breast cancer patients younger than 40 years. 55: 1578-1583
(78) Radiology 2015; Hyun SH, et al. Volume-based metabolic tumor response to neoadjuvant chemotherapy is associated with an increased risk of recurrence in breast cancer. 275: 235-244
(79) J Nucl Med 2015; Champion L, et al. 18F-FDG PET/CT to predict response to neoadjuvant chemotherapy and prognosis in inflammatory breast cancer. 56: 1315-1321
(80) AJR 2015; Son SH, et al. Whole-body metabolic tumor volume, as determined by 18F-FDG/PET/CT, as a prognsotic factor of outcome for patients with breast cancer who have distant metastases. 205: 878-885
(81) AJR 2015; Jochelson MS, et al. Detection of internal mammary adenopathy in patients with breast cancer by PET/CT and MRI. 205: 899-904
(82) Radiology 2015; Groheux D, et al. Early metabolic response to neoadjuvant treatment: FDG PET/CT criteria according to breast cancer subtype. 277: 358-371
(83) Radiology 2015; Sutton EJ, et al. Incidence of internal mammary lymph nodes with silicone breast implants at MR imaging after oncoplastic surgery. 277: 381-387
(84) J Nucl Med 2015; Hogan MP, et al. Comparison of 18F-FDG PET/CT for systemic staging of newly diagnosed invasive lobular carcinoma versus invasive ductal carcinoma. 56: 1674-1680
(85) J Nucl Med 2015; Bae MS, et al. Early stage triple-negative breast cancer: imaging and clinical-pathologic factors associated with recurrence. 278: 356-364
(86) J Nucl Med 2016; Groheux D, et al. 18F-FDG PET/CT for staging and restaging of breast cancer. 57: 17S-26S
(87) J Natl Cancer Inst 2012; Groheux D, et al. Prognostic impact of 18F-FDG PET-CT findings in clinical stage III and IIB breast cancer. 104: 1879-87
(88) J Nucl Med 2016; Cook GJR, et al. Imaging bone metastases in breast cancer: staging and response assessment. 57: 27S-33S
(89) J Nucl Med 2016; Avril S, et al. 18F-FDG PET/CT for monitoring of treatment response in breast cancer. 57: 34S-39S
(90) AJR 2016; Taghipour M, et al. FDG avidity and tumor burden: survival outcomes for patients with recurrent breast cancer. 206: 846-855
(91) J Nucl Med 2016; Lee HW, et al. The prognostic impact of early change in 18F-FDG PET SUV after neoadjuvant chemotherapy in patients with locally advanced breast cancer. 57: 1183-1188
(92) J Nucl Med 2016; Liao GJ, et al. 18F-fluoroestradiol PET: current status and potential future clinical applications. 56: 1269-1275
(93) AJR 2017; Rauch GM, et al. Multimodality imaging for evaluating response to neoadjuvant chemotherapy in breast cancer. 208: 290-299
(94) J Nucl Med 2017; Lebon V, et al. Rate of distant metastases on 18F-FDG PET/CT at initial staging of breast cancer: comparison of women younger and older than 40 years. 58: 252-257
(95) J Nucl Med 2017; Fowler AM, Linden HM. Functional estrogen receptor imaging before neoadjuvant therapy for primary breast cancer. 58: 560-562
(96) J Nucl Med 2017; Chae SY, et al. A randomized feasibility study of 18F-fluoroestradiol PET to predict pathologic response to neoadjuvant therapy in estrogen receptor-rish postmenopausal breast cancer. 58: 563-568
(97) Radiology 2017; Cox VL, et al Cancer genomics and important mutations: a contemporary guide for body imagers. 283: 314-340
(98) Radiographics 2017; Lee MV, et al. BRCA-associated cancers: role of imaging in screening, diagnosis, and management. 37: 1005-1023
(99) AJR 2017; Ha SM, et al. Association of BRCA mutation types, imaging features, and pathologic findings in patients with breast cancer with BRCA1 and BRCA2 mutations. 209: 920-928
(100) J Nucl Med 2018; Nienhuis HH, et al. 18F-fluoroestradiol tumor uptake is heterogeneous and influenced by site of metastasis in breast cancer. 59: 1212-1218
(101) AJR 2019; Satoh Y, et al. Unexpected abnormal uptake in the breasts at dedicated breast PET: incidentally detected small cancers or nonmalignant features? 212: 443-449
(102) AJR 2019; Ulaner GA. PET/CT for patients with breast cancer: where is the clinical impact? 213: 254-265
(107) J Nucl Med 2021; Peterson LM, et al. 18F-fluoroestradiol PET imaging in a phase II trial of Vorinostat to restore endocrine sensitivity in ER+/HER2- metastatic breast cancer. 62: 184-190
(109) J Nucl Med 2021; Boers J, et al. Value of 18F-FES PET in solving clinical dilemmas in breast cancer patients: a retrospective study. 62: 1214-1220
(111) Radiology 2022; Mankoff DA, Sellmeyer MA. PET of fibroblast-activation protein for breast cancer diagnosis and staging. 302: 48-49
(112) Radiographics 2022; Gao Y, et al. Non-BRCA early-onset breast cancer in young women. 42: 5-22
(116) J Nucl Med 2023; Ulaner GA, et al. Summary: appropriate use criteria for estrogen receptor-targeted PET imaging with 16-alpha 18F-fluoro-17beta-fluoroestradiol. 64: 351-354
(117) J Nucl Med 2023; Vogsen M, et al. Response monitoring in metastatic breast cancer: a prospective study comparing 18F-FDG PET/CT with conventional CT. 64: 355-361
(118) Radiographics 2023; 18F-fluoroestradiol: current applications and future directions. 43: (e220143) 1-17
(119) AJR 2023; Covington MF, et al. Prospective pilot study of 18F-fluoroestradiol PET/CT in patients with invasive lobular carcinomas. 211: 228-239
(121) J Nucl Med 2023; Gebhart G. 18F-FDG PET "metabolic response" to neoadjuvant therapy for breast cancer: quo vadis? 64: 1697-1698