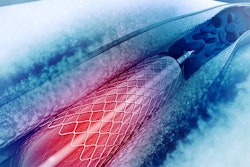
Royal Philips, the parent company of Philips Healthcare, has relaunched its Pioneer Plus catheter, a re-entry device with intravascular ultrasound (IVUS) capabilities and needle deployment designed to assist arterial vessel intervention.
Vessels in the peripheral artery system are captured using IVUS, which allows physicians to identify a targeted area and place a catheter in a specific location. The catheter also provides a cross-sectional ultrasound image of the arterial area that allows for guidewire placement beyond stenotic lesions prior to additional interventions.
Philips initiated a design modification to the Pioneer Plus catheter in 2015, prompting the company to take the product off the market proactively until completing modifications; it has now reissued the catheter because the modifications are finished. The catheter has U.S. Food and Drug Administration (FDA) clearance and CE Mark approval, according to the vendor.