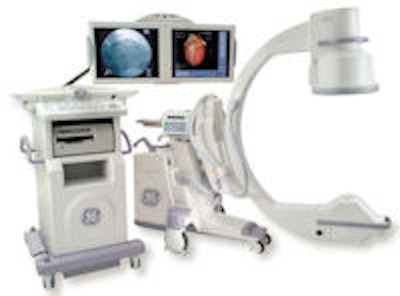
Multimodality vendor GE Healthcare of Chalfont St. Giles, U.K., has received the go-ahead from the U.S. Food and Drug Administration (FDA) to begin manufacturing and distributing surgical C-arm shipments from its GE Healthcare Surgery subsidiary. The vendor plans to resume shipments pending the receipt of 510(k) clearance of the first product to be manufactured under new processes the company has put in place.
On May 1, the FDA notified GE that the company had met the requirements of a consent decree between the parties that spelled out the terms under which GE could resume shipping C-arms from two Healthcare Surgery facilities. GE suspended C-arm manufacturing and distribution in August 2006 after an FDA inspection of facilities in Salt Lake City and Lawrence, MA.
GE has been retooling its good manufacturing practices (GMPs) in response to the FDA's concerns. The company said it will begin shipments as soon as it receives 510(k) clearance for the OEC 9900 Elite C-arm, and the vendor expects to ship 300 units in the first 10 days after receiving the FDA's go-ahead, according to Pete McCabe, president and CEO of GE OEC Medical Systems.
The FDA's move is in response to several key steps that have been completed as part of GE's effort to implement the terms of the consent decree. First, GE completed a corrective action plan for fixing installed C-arms in the field. The company then had a third-party firm inspect and certify the company's quality management system for its manufacturing operations. Finally, the FDA itself inspected GE Healthcare Surgery facilities and signed off on the company's progress.
GE decided to file a new 510(k) submission for the 9900 Elite C-arm because the company felt it had made enough modifications to the system to warrant a new application, McCabe said. GE tackled the 9900 first because that system is the company's flagship C-arm; GE will also go through the same process for the 9800 model, though McCabe said the company isn't yet sure whether it will have to file a 510(k) for that system.
Many of the changes made to the 9900 C-arm are relatively minor, such as fixing software bugs that may have caused the system to boot up improperly, or adding an uninterruptible power supply to the system so that the last image on the unit is still available even if the system is unplugged from its power supply. McCabe believes that its C-arms are now twice as reliable as they were previously.
McCabe estimates that the suspension cost GE hundreds of millions of dollars in lost revenues and in expenses related to changes in its manufacturing processes. However, he believes that in the end, the company is better for the experience.
"We are a much better business for going through this process," McCabe said. "We hate the way we had to get here, but we're a better business."
By Brian Casey
AuntMinnie.com staff writer
May 2, 2008
Related Reading
GE to build x-ray facility in Brazil, May 1, 2008
GE in Saudi Arabian joint venture, April 28, 2008
DeJarnette teams with GE, April 24, 2008
GE signs the University of Tennessee, April 17, 2008
GE Healthcare reports tough Q1, April 11, 2008
Copyright © 2008 AuntMinnie.com