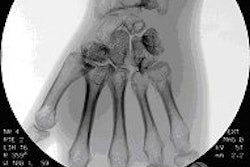
Imaging equipment developer Analogic of Peabody, MA, has received Food and Drug Administration 510(k) clearance for its new multipurpose digital radiography system, SyneRad Omni.
The device features the GR17 amorphous selenium flat-panel detector from Analogic's Anrad subsidiary in Canada, and received 510(k) clearance earlier this month, the company said. SyneRad Omni provides multiframe acquisition capability in full 17 x 17-inch fields, allowing it to provide expanded clinical utility in areas such as joint stability and motion studies.
The product is being sold by Analogic subsidiary Anexa, and will be exhibited at the RSNA conference in Chicago next week.
By AuntMinnie.com staff writers
November 24, 2004
Related Reading
Road to RSNA, Anexa, November 11, 2004
Vanderbilt picks Anexa for DR, November 4, 2004
Analogic selects Burch as VP, October 1, 2004
Analogic Q4 revenues surge, September 22, 2004
Analogic taps Harris as security VP, September 16, 2004
Copyright © 2004 AuntMinnie.com